BLOG List
Shelf Life Test Chamber for Temperature and Humidity Cosmetic Testing
Imagine your cosmetic or food product quietly aging on a store shelf—enduring summer heat, winter cold, and fluctuating humidity long before it ever reaches the consumer. A shelf life test chamber makes this invisible journey visible, simulating real-world storage conditions to uncover formulation weaknesses, packaging risks, and long-term stability challenges before market launch.
This performance is already being validated in real laboratories. A Canadian lab using the TH-1000C shelf life test chamber shared early feedback: “Hi Karen, we have been using the chamber since last week. It is overall good so far. I will keep you updated.” Such real-world confidence highlights the reliability and consistency required for cosmetic stability testing. Let’s explore how shelf life test chambers safeguard product quality, safety, and brand trust.
A shelf life test chamber designed for cosmetic testing simulates real-world storage conditions to evaluate product stability over time. These specialized environmental chambers control temperature (-40°C to +150°C) and humidity (20%-98% RH) to replicate months or years of aging within weeks. By exposing cosmetics to accelerated conditions, manufacturers can predict shelf life, identify formulation weaknesses, verify packaging integrity, and ensure regulatory compliance before market launch. This testing validates that products maintain their safety, efficacy, and aesthetic qualities throughout their intended lifespan, protecting both brand reputation and consumer trust.
Why Cosmetics Require Stability Testing?
Regulatory Compliance Requirements
Cosmetic manufacturers worldwide face stringent regulatory frameworks that mandate stability testing before product distribution. The FDA's Cosmetic Voluntary Registrations Program and the EU's Cosmetics Regulation (EC) No 1223/2009 require documented evidence of product safety and quality maintenance. A shelf life test chamber provides the controlled environment necessary to generate this compliance data, demonstrating that formulations remain within acceptable specifications throughout their declared period after opening (PAO). Testing protocols typically span 12-36 months under various stress conditions, with chambers providing reproducible results that satisfy regulatory auditors.
Registrations Program and the EU's Cosmetics Regulation (EC) No 1223/2009 require documented evidence of product safety and quality maintenance. A shelf life test chamber provides the controlled environment necessary to generate this compliance data, demonstrating that formulations remain within acceptable specifications throughout their declared period after opening (PAO). Testing protocols typically span 12-36 months under various stress conditions, with chambers providing reproducible results that satisfy regulatory auditors.
Consumer Safety Considerations
Degraded cosmetics pose significant health risks ranging from skin irritation to microbial contamination. Temperature and humidity fluctuations accelerate chemical reactions that can transform safe ingredients into sensitizing compounds. Preservative systems may lose efficacy, allowing bacterial or fungal growth that threatens user safety. Environmental simulation chambers expose products to worst-case scenarios - tropical heat, freezing winters, humid coastal climates - revealing vulnerabilities before consumers encounter them. This proactive approach prevents recalls, liability claims, and reputational damage while safeguarding public health.
Market Competition and Brand Protection
Premium cosmetic brands differentiate themselves through guaranteed product performance across global markets. Stability testing validates claims about texture retention, fragrance longevity, and color consistency that justify higher price points. Chambers enable comparative testing against competitor formulations, identifying competitive advantages or necessary improvements. Documented stability data becomes a marketing asset, supporting claims of superior quality and providing technical ammunition for B2B negotiations with retailers demanding shelf life guarantees.
Effects of Heat and Moisture on Cosmetic ProductsChemical Degradation Pathways
Elevated temperatures accelerate oxidative reactions that destabilize active ingredients and cause color shifts. Retinol converts to inactive isomers, vitamin C degrades to dehydroascorbic acid, and essential oils lose volatile components. Humidity introduces water activity that hydrolyzes esters, disrupts emulsions, and enables Maillard browning reactions. A controlled shelf life test chamber replicates these conditions with precision, using PT100Ω temperature probes and humidity sensors to maintain exact parameters. The programmable touchscreen controller cycles between stress conditions, simulating diurnal temperature variations and seasonal humidity changes that reveal formulation resilience.
Physical Stability Changes
Heat reduces viscosity in creams and lotions, causing phase separation where oil and water components divorce. Cold temperatures induce crystallization in lipid-based products, creating grainy textures. Humidity affects powder compacts through moisture absorption, causing clumping or microbial growth. Environmental chambers equipped with forced air convection systems ensure uniform conditions throughout the testing space, eliminating hot spots that could produce misleading results. The mirror-finished stainless steel interiors reflect heat evenly while simplifying cleanup after inevitable product spillage.
Microbial Challenge Response
Temperature and humidity directly influence preservative efficacy and microbial survival rates. Chambers operating at 30°C/75% RH replicate tropical conditions where bacteria and fungi thrive, challenging antimicrobial systems to their limits. Conversely, freeze-thaw cycling tests packaging seal integrity and preservative distribution after temperature shock. Multi-cycle testing protocols in advanced chambers reveal whether preservative systems maintain minimum inhibitory concentrations (MIC) against common cosmetic contaminants like Pseudomonas aeruginosa and Candida albicans throughout the product lifecycle.
| Name | shelf life test chamber | |||||
|
Model |
TH-100 |
|||||
|
Temperature range |
-20℃ ~+150 ℃ |
|||||
|
Low type |
A: -40℃ B:-70℃ C -86℃ |
|||||
|
Humidity Range |
20%-98%RH |
|||||
|
Temperature deviation |
± 2.0 ℃ |
|||||
|
Heating rate |
3 ℃ / min |
|||||
|
Cooling rate |
1 ℃ / min |
|||||
|
Controller |
Programmable color LCD touch screen controller, Multi-language interface, Ethernet , USB |
|||||
|
Exterior material |
Steel Plate with protective coating |
|||||
|
Interior material |
SUS304 stainless steel |
|||||
|
Standard configuration |
1 Cable hole (Φ 50) with plug; 2 shelves |
|||||
|
Timing Function |
0.1~999.9 (S,M,H) settable |
|||||
Packaging Evaluation for Cosmetic Shelf LifeContainer Material Compatibility
Packaging materials respond dynamically to environmental stresses, potentially contaminating products or failing structurally. Plastic containers may leach plasticizers at elevated temperatures, while metals can corrode under humid conditions. Glass shows minimal reactivity but risks thermal shock cracking. Shelf life chambers with temperature ranges from -86°C to +150°C test packaging across extreme conditions, revealing material limitations before mass production commitments. Sample shelves constructed from perforated #304 stainless steel accommodate various container sizes while ensuring air circulation around each test specimen.
Seal Integrity and Barrier Properties
Moisture vapor transmission rates (MVTR) and oxygen permeability determine how quickly external conditions affect product quality. Chamber testing quantifies these barrier properties under stress, measuring weight changes that indicate moisture ingress or volatile loss. Airless pumps, droppers, and spray mechanisms undergo functional testing after temperature cycling to ensure dispensing accuracy. The TH-225 and TH-500 models offer sufficient internal dimensions (500×600×750mm and 700×800×900mm respectively) to accommodate full product packaging rather than just formulation samples.
|
Parameter |
TH-225 Model |
TH-500 Model |
|
Internal Dimensions |
500×600×750 mm |
700×800×900 mm |
|
Temperature Range |
-86°C to +150°C |
-86°C to +150°C |
|
Humidity Range |
20%-98% RH |
20%-98% RH |
|
Temperature Deviation |
±2.0°C |
±2.0°C |
Label and Print Durability
Product labeling conveys critical safety information that must remain legible throughout shelf life. Humidity causes adhesive failure, ink bleeding, and paper delamination. UV exposure fades colors while heat accelerates adhesive degradation. Shelf life test chambers simulate these combined stresses, revealing whether regulatory warnings, ingredient lists, and brand graphics survive realistic storage and distribution scenarios. Testing prevents marketplace failures where illegible labels trigger regulatory violations or consumer confusion.
Accelerated Aging Methods in CosmeticsQ10 Temperature Coefficient Calculations
The Arrhenius equation provides the mathematical foundation for accelerated aging, where each 10°C temperature increase approximately doubles reaction rates (Q10 ≈ 2). Chambers operating at 40°C simulate roughly 6-12 months of room temperature aging per month of testing, depending on specific reaction kinetics. Precise temperature control within ±0.5°C fluctuation ensures reliable extrapolation from accelerated to real-time conditions. Programmable controllers execute complex temperature profiles that alternate stress periods with recovery intervals, mimicking seasonal variations more accurately than constant-temperature protocols.
ICH Stability Testing Guidelines
The International Council for Harmonisation (ICH) establishes globally recognized testing conditions including 25°C/60% RH (Zone II, temperate climates) and 30°C/65% RH (Zone IVa, hot/humid climates). Advanced shelf life chambers store multiple ICH protocols simultaneously, with independent compartments maintaining different conditions. The environmentally friendly R404A and R23 refrigerants provide efficient cooling across the -86°C to +150°C operational range while complying with environmental regulations. Ethernet connectivity allows remote monitoring of multiple ongoing studies, with automatic alerts when parameters drift beyond specifications.
Photostability Testing Integration
While traditional chambers focus on temperature and humidity, comprehensive stability programs include light exposure simulating retail display conditions. Some customized units integrate UV/visible light sources alongside climate control, enabling simultaneous photo-degradation and thermal stress testing. The USB data logging capability captures minute-by-minute conditions, providing the documentation trail regulators demand. Multi-language touchscreen interfaces accommodate international research teams collaborating on global product launches.
|
Testing Condition |
Climate Zone |
Typical Duration |
Purpose |
|
25°C / 60% RH |
Zone II (Temperate) |
12-36 months |
Standard shelf life validation |
|
30°C / 65% RH |
Zone IVa (Hot/Humid) |
6-12 months |
Tropical market qualification |
|
40°C / 75% RH |
Accelerated |
3-6 months |
Challenge testing |
Ensuring Product Safety and PerformanceActive Ingredient Potency Maintenance
Cosmeceutical products containing pharmaceutical-grade actives require validated stability proving therapeutic concentrations persist until expiration. Peptides undergo hydrolysis, antioxidants sacrifice themselves neutralizing free radicals, and enzymes denature under stress. Shelf life test chamber testing with regular sampling intervals (0, 1, 3, 6, 12 months) generates degradation curves showing potency over time. Analytical methods like HPLC quantify remaining active concentrations, determining whether products maintain minimum effective doses throughout their claimed shelf life. This data supports labeling accuracy and therapeutic claims.
undergo hydrolysis, antioxidants sacrifice themselves neutralizing free radicals, and enzymes denature under stress. Shelf life test chamber testing with regular sampling intervals (0, 1, 3, 6, 12 months) generates degradation curves showing potency over time. Analytical methods like HPLC quantify remaining active concentrations, determining whether products maintain minimum effective doses throughout their claimed shelf life. This data supports labeling accuracy and therapeutic claims.
Sensory Attribute Preservation
Consumer acceptance depends heavily on sensory experiences - fragrance, color, texture, and application feel. Heat accelerates fragrance note development and evaporation, shifting top notes to base notes prematurely. Emulsion breakdown produces separated layers or grainy textures. Color changes from oxidation signal quality degradation even when products remain safe. Trained sensory panels evaluate chamber-aged samples against fresh controls, quantifying perceptible differences using standardized scoring systems. Products failing sensory benchmarks before expiration dates require reformulation regardless of safety test passage.
Consistency Across Production Batches
Stability testing isn't merely a development activity - ongoing quality assurance programs test representative samples from each production batch. Chamber archives retain reserve samples from every batch, enabling retrospective investigation when customer complaints arise. The independent water and electrical systems in LIB chambers ensure continuous operation even during maintenance, preventing testing interruptions that could delay product releases. Remote control capabilities via laboratory networks allow quality managers to monitor multiple chambers across different facilities simultaneously.
Quality Control Metrics for Cosmetic StabilityPhysical Property Measurements
Objective testing protocols measure viscosity changes, pH drift, specific gravity variations, and particle size distributions. Rheological testing reveals how creams and lotions flow and spread, critical parameters affecting consumer perception and application effectiveness. pH monitoring detects formulation instability or preservative breakdown, with acceptable ranges typically within ±0.5 pH units of initial values. Color spectrophotometers quantify Lab values, detecting subtle shifts invisible to untrained observers. Chambers provide the stable baseline conditions necessary for reproducible measurements across testing timepoints.
Microbiological Stability Indicators
Preservative challenge testing exposes aged samples to standardized microbial inocula (bacteria, yeast, mold) to verify antimicrobial system functionality. Pass criteria require specific log reductions within defined timeframes - typically 2-log reduction within 14 days for bacteria, 1-log for fungi. Total viable count (TVC) testing throughout stability studies detects contamination from inadequate preservation or packaging failures. The easy-to-clean mirror stainless steel interiors with punch-hole drainage design prevent microbial reservoirs within chambers that could cross-contaminate test samples.
Analytical Chemistry Validation
Gas chromatography, mass spectrometry, and spectroscopic methods identify degradation products, preservative concentrations, and fragrance component levels. Stability-indicating assays distinguish active ingredients from degradation products, quantifying both desired compounds and potentially harmful breakdown products. Acceptance criteria established during development define acceptable ranges for each measured parameter. Chamber-generated samples feed systematic analytical programs producing the documentation chains regulatory agencies scrutinize during pre-market reviews and post-market surveillance.
|
Quality Metric |
Testing Method |
Acceptance Criteria |
Frequency |
|
Viscosity |
Brookfield Rheometer |
±20% of initial value |
0, 3, 6, 12 months |
|
pH Level |
Electrode Measurement |
±0.5 pH units |
0, 3, 6, 12 months |
|
Color Stability |
Spectrophotometry |
ΔE < 2.0 |
0, 3, 6, 12 months |
|
Preservative Content |
HPLC Analysis |
>80% of label claim |
0, 6, 12 months |
Preserve Cosmetic Integrity with LIB Industry's Controlled Shelf Life Test ChamberAdvanced Technical Specifications
LIB Environmental Simulation Industry delivers shelf life test chambers combining precision control with operational reliability. The forced air convection system produces uniform temperature distribution eliminating dead zones that compromise test validity. High-precision PT100Ω sensors monitor conditions with minimal drift, maintaining ±2.0°C temperature deviation across the entire workspace. Heating rates of 3°C/min and cooling rates of 1°C/min enable efficient protocol cycling without thermal overshoot. The programmable color LCD touchscreen controller accommodates complex multi-stage protocols with intuitive menu navigation, reducing operator training requirements.
Customization and Integration Capabilities
LIB's engineering team configures chambers matching specific testing requirements through extensive customization options. Explosion-proof modifications accommodate volatile formulations, low-pressure simulation replicates high-altitude distribution environments, and integrated vibration systems combine transport stress with climate exposure. The standard network ports enable seamless integration into laboratory information management systems (LIMS), automatically logging data and triggering alerts when parameters deviate. Water level observation windows and independent water/electrical system architecture enhance safety while simplifying maintenance. Multiple refrigerant options (-40°C, -70°C, -86°C low-temperature configurations) serve diverse testing needs from standard cosmetics to specialty products requiring extreme cold exposure.
Comprehensive Support and Validation
Beyond equipment manufacturing, LIB provides turnkey solutions encompassing installation, commissioning, operator training, and ongoing technical support. CE certification demonstrates compliance with European safety standards, while factory acceptance testing (FAT) and site acceptance testing (SAT) validate performance before customer acceptance. Calibration certificates traceable to national standards accompany each chamber, establishing measurement confidence for regulatory submissions. Extended service contracts include preventive maintenance, calibration verification, and emergency response, ensuring continuous operation supporting critical stability programs. The global support network delivers responsive technical assistance regardless of installation location, protecting research investments and project timelines.
Conclusion
Shelf life test chambers represent indispensable infrastructure for cosmetic manufacturers committed to quality, safety, and regulatory compliance. By simulating diverse environmental conditions with precision control, these systems predict product performance across global markets and storage scenarios. The investment in validated stability testing prevents costly recalls, protects brand reputation, and demonstrates commitment to consumer safety. LIB Industry's comprehensive chamber solutions deliver the technical capabilities, customization flexibility, and ongoing support necessary for rigorous cosmetic stability programs meeting international standards.
FAQsHow long does cosmetic stability testing typically require in a shelf life test chamber?
Standard testing protocols run 12-36 months for real-time studies, while accelerated testing at elevated temperatures compresses this timeline to 3-6 months. The specific duration depends on intended shelf life claims, regulatory requirements, and product complexity. Multiple timepoint sampling throughout testing generates comprehensive degradation profiles.
What temperature and humidity conditions should cosmetics be tested under?
ICH guidelines recommend 25°C/60% RH for temperate climates and 30°C/65% RH for hot/humid regions as primary conditions. Accelerated testing typically employs 40°C/75% RH, while stress testing may include freeze-thaw cycling and extreme temperatures up to 50°C. Testing should reflect the harshest conditions expected during distribution and storage.
Can one chamber test multiple product formulations simultaneously?
Yes, shelf life chambers accommodate numerous samples simultaneously through adjustable shelving systems. However, all samples within a chamber experience identical conditions. Testing products requiring different climate protocols necessitates multiple chambers or sequential testing programs. Sample placement should ensure adequate air circulation and avoid cross-contamination between formulations.
Partner with LIB Industry, a leading shelf life test chamber manufacturer and supplier, to ensure your cosmetic products maintain integrity throughout their lifecycle. Contact our technical team at ellen@lib-industry.com to discuss customized testing solutions for your specific formulation and packaging requirements.
Using a UV Resistance Test Chamber for Coatings Weathering Evaluation
Coating weathering evaluation through UV resistance test chambers enables manufacturers to predict material performance before real-world deployment. These specialized chambers replicate ultraviolet radiation, temperature fluctuations, and moisture conditions that cause coating degradation outdoors. By accelerating natural weathering processes, laboratories can assess gloss retention, color stability, and surface integrity within weeks rather than years. This controlled methodology helps coating formulators optimize product formulations, validates protective performance claims, and ensures compliance with international durability standards across automotive, architectural, and industrial applications.
Why Weathering Testing is Essential for Coatings?Protecting Investment and Brand Reputation
Coatings serve as the primary defense mechanism against environmental degradation for countless products and structures. When protective layers fail prematurely, manufacturers face warranty claims, customer dissatisfaction, and potential safety hazards. Weathering evaluation identifies formulation weaknesses before market release, preventing costly field failures. Automotive manufacturers, architectural firms, and industrial equipment suppliers rely on accelerated testing to validate performance claims and maintain competitive positioning.
warranty claims, customer dissatisfaction, and potential safety hazards. Weathering evaluation identifies formulation weaknesses before market release, preventing costly field failures. Automotive manufacturers, architectural firms, and industrial equipment suppliers rely on accelerated testing to validate performance claims and maintain competitive positioning.
Understanding Real-World Degradation Mechanisms
Natural weathering involves complex interactions between UV radiation, moisture, temperature cycles, and atmospheric pollutants. These factors trigger photochemical reactions, hydrolysis, thermal expansion, and oxidative stress within coating matrices. Laboratory testing isolates and intensifies these variables, revealing degradation pathways that inform formulation improvements. Researchers can systematically evaluate how different resin systems, pigments, and additives respond to environmental stressors.
Meeting Customer Expectations for Durability
End-users increasingly demand extended service life from coated products, whether architectural facades lasting decades or automotive finishes maintaining appearance through years of exposure. Weathering data provides quantifiable evidence of durability, supporting marketing claims and technical specifications. This transparency builds customer confidence and differentiates premium products in competitive markets.
Simulating Sunlight and Environmental Stress on CoatingsReplicating UV Spectral Distribution
Natural sunlight contains UV radiation spanning 290-400 nanometers, with different wavelengths causing distinct degradation mechanisms. UVA radiation (340nm) drives long-term photochemical reactions, while UVB (313nm) produces more aggressive surface damage. Modern UV resistance test chambers utilize specialized fluorescent lamps calibrated to specific wavelengths, matching outdoor exposure conditions for particular geographic locations or application environments.
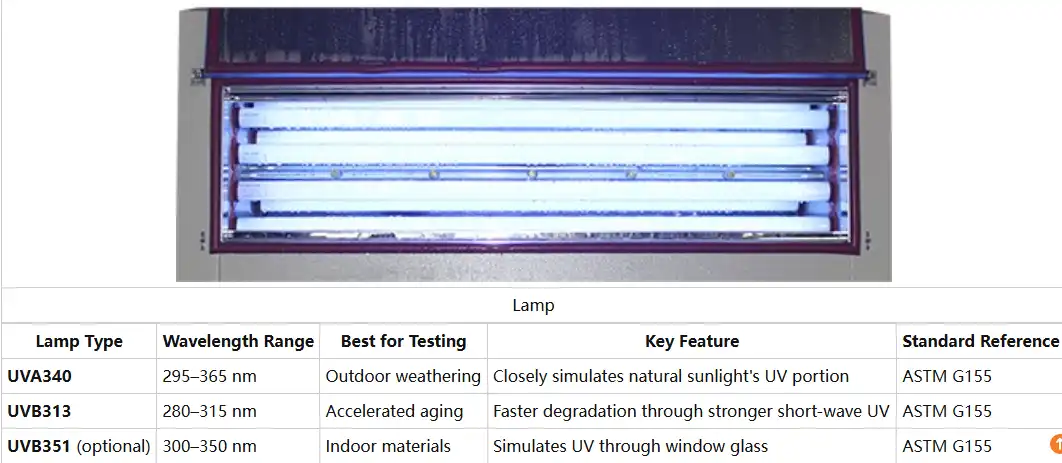
Incorporating Moisture and Temperature Cycling
Coating degradation accelerates when UV exposure combines with moisture presence and thermal stress. Test chambers integrate water spray systems that simulate dew, rain, and humidity conditions. Programmable controllers cycle between UV irradiation phases and moisture exposure, replicating daily weather patterns. Temperature control systems maintain precise conditions between ambient and 90°C, inducing thermal expansion and contraction that stresses coating-substrate interfaces.

Controlling Irradiance Levels for Consistency
UV intensity varies with season, latitude, and atmospheric conditions in natural environments. Laboratory chambers maintain constant irradiance between 0.3-20 W/m², ensuring reproducible test conditions across different facilities and time periods. This precision enables comparative evaluation of coating formulations and facilitates correlation between accelerated test results and outdoor exposure data from reference sites.
Evaluating Color, Gloss, and Surface Integrity
Quantifying Color Stability and Fade Resistance
Color changes represent the most visible weathering symptom, directly impacting aesthetic appeal and customer satisfaction. Spectrophotometers measure color coordinates in standardized color spaces (Lab or LCh), calculating total color change (ΔE) after specified exposure intervals. Coating formulators target ΔE values below acceptable thresholds, typically ranging from 2-5 units depending on application requirements. Pigment selection, dispersant chemistry, and stabilizer packages significantly influence fade resistance.
standardized color spaces (Lab or LCh), calculating total color change (ΔE) after specified exposure intervals. Coating formulators target ΔE values below acceptable thresholds, typically ranging from 2-5 units depending on application requirements. Pigment selection, dispersant chemistry, and stabilizer packages significantly influence fade resistance.
Monitoring Gloss Retention Through Exposure
Surface gloss deteriorates as UV radiation degrades resin networks and creates microscopic surface roughness. Glossmeters positioned at 20°, 60°, or 85° angles measure specular reflectance, tracking gradual reduction through weathering cycles. High-performance architectural coatings maintain 80% initial gloss after 2000 hours accelerated exposure, while automotive clearcoats require even stricter retention. Gloss loss correlates with consumer perception of product aging and quality.
|
Performance Level |
Gloss Retention @ 2000h |
Typical Applications |
|
Premium |
>85% |
Automotive OEM finishes |
|
Standard |
70-85% |
Architectural coatings |
|
Economy |
50-70% |
Industrial maintenance |
Assessing Surface Defects and Mechanical Properties
Advanced weathering produces chalking, cracking, blistering, and delamination - defects compromising both appearance and protective function. Visual inspection following standardized rating scales (ASTM methods) documents surface deterioration progression. Complementary mechanical testing evaluates flexibility, adhesion, and hardness changes. Coating systems must maintain critical performance thresholds throughout intended service life, requiring iterative formulation optimization validated through accelerated testing.
Accelerated UV Exposure Techniques for CoatingsUnderstanding Acceleration Factors
Accelerated weathering compresses years of outdoor exposure into weeks or months by intensifying UV irradiance and optimizing exposure conditions. Acceleration factors ranging from 4:1 to 12:1 are achievable depending on lamp selection, irradiance levels, and cycling protocols. However, excessive acceleration risks altering degradation mechanisms, potentially producing unrealistic failure modes. Correlation studies comparing UV resistance test chambers results with outdoor exposure sites validate acceleration factors for specific coating types.
Designing Exposure Cycles for Realistic Results
Optimal test protocols alternate UV exposure with moisture condensation phases, mimicking natural diurnal cycles. A typical automotive coating protocol might specify 8 hours UV at 60°C followed by 4 hours condensation at 50°C, repeated continuously. Seasonal variations can be simulated through longer-term protocols incorporating intensity adjustments. Protocol selection depends on intended application environment - tropical, temperate, or arid conditions require different cycling patterns.
Comparing Chamber Testing with Outdoor Exposure
Outdoor weathering stations in locations like Florida, Arizona, and Australia provide real-world validation for accelerated test predictions. Side-by-side exposure programs track coating performance both in chambers and outdoor racks, establishing correlation equations. This dual approach confirms that laboratory findings accurately predict field performance while maintaining testing efficiency. Discrepancies prompt protocol refinement to improve predictive accuracy.
Predicting Coating Lifespan under Harsh ConditionsEstablishing Performance Endpoints
Service life predictions require clearly defined failure criteria based on application requirements. Architectural coatings might specify maximum acceptable color change, minimum gloss retention, and absence of cracking. Automotive finishes incorporate stricter aesthetic thresholds alongside functional requirements. Test protocols incorporate intermediate measurements at regular intervals, plotting performance degradation curves that indicate when failure criteria will be exceeded.
Extrapolating Laboratory Data to Real-World Conditions
Mathematical models translate accelerated test results into outdoor exposure equivalents using reciprocity principles and dose-response relationships. Cumulative UV dose received in chambers correlates with annual outdoor exposure in specific climates. Weathering databases containing decades of outdoor exposure data support these extrapolations. Statistical analysis incorporates confidence intervals, acknowledging uncertainties inherent in accelerated testing predictions.
Validating Durability Claims for Warranties
Manufacturers rely on weathering data to substantiate warranty periods and performance guarantees. A coating warranted for 10-year durability must demonstrate adequate performance when subjected to equivalent accelerated exposure. Regulatory bodies and industry associations increasingly require documented weathering evidence supporting marketing claims. This validation protects manufacturers from unsubstantiated claims while providing customers with reliable performance expectations.
Ensuring Compliance with Industry StandardsNavigating ASTM and ISO Testing Standards
Standardized test methods ensure reproducibility and enable industry-wide performance comparisons. ASTM G154 specifies fluorescent UV exposure procedures, detailing lamp types, irradiance levels, and exposure cycles. ISO 4892 provides parallel guidance recognized internationally. Additional standards address specific coating types - ASTM D7356 covers architectural coatings, while SAE J2527 focuses on automotive applications. Compliance with relevant standards facilitates product qualification and customer acceptance.
Meeting Automotive and Architectural Specifications
Original equipment manufacturers maintain proprietary weathering specifications exceeding generic standards. Automotive companies require extensive testing programs spanning thousands of hours with multiple evaluation checkpoints. Architectural coating specifications from organizations like AAMA (American Architectural Manufacturers Association) define performance classes based on weathering resistance. Suppliers must demonstrate compliance through certified testing facilities using UV resistance test chambers.
|
Standard |
Application |
Key Requirements |
|
ASTM G154 |
General UV weathering |
Lamp type, irradiance, cycling protocol |
|
ISO 4892-3 |
International weathering |
Fluorescent UV methods and procedures |
|
AAMA 2605 |
Architectural coatings |
4000h exposure with color/gloss limits |
|
SAE J2527 |
Automotive exterior |
2000h+ with appearance retention |
Documenting Test Results for Regulatory Approval
Comprehensive test reports document equipment calibration, exposure conditions, measurement procedures, and performance results. Chain-of-custody protocols and laboratory accreditation ensure data integrity for regulatory submissions. Regulatory agencies reviewing coating approvals for food contact, medical devices, or hazardous environments scrutinize weathering evidence demonstrating maintained protective properties. Thorough documentation expedites approval processes and withstands regulatory scrutiny.
Accelerated Aging Studies with LIB Industry's UV Resistance Test ChamberTechnical Capabilities Supporting Research Programs
The LIB UV-SI-260 chamber provides controlled weathering environments accommodating 56 specimens simultaneously. Eight 40-watt fluorescent UV lamps deliver precisely controlled irradiance between 0.3-20 W/m² across the 290-400nm bandwidth. Temperature regulation spanning ambient to 90°C with ±2°C precision ensures consistent exposure conditions. Programmable color LCD touch screen controllers enable complex cycling protocols with independent control of UV, temperature, and moisture phases lasting up to 9999 hours.
Flexible Sample Configuration and Monitoring
Removable sample holders accommodate both flat and three-dimensional specimens measuring 75×150mm, allowing evaluation of complex geometries. The 50mm lamp-to-specimen distance ensures uniform irradiance distribution across the exposure area. Automated water spray systems with ten 0.5mm nozzles deliver controlled moisture application at 0.8-1.0mm per 80cm² hourly. Black panel temperature monitoring provides additional exposure condition verification, measuring actual specimen surface temperatures during testing.
Supporting Quality Assurance and Product Development
Research laboratories utilize the chamber for formulation screening, comparing multiple candidate coatings under identical conditions. Quality control departments validate production batch consistency against reference standards. Product development teams optimize additive packages and resin selection based on systematic weathering evaluations. The chamber's Ethernet connectivity enables remote monitoring and data logging, supporting modern laboratory information management systems and quality documentation requirements.
Conclusion
UV resistance test chambers provide indispensable tools for modern coating development and quality assurance programs. By replicating years of environmental exposure within controlled laboratory timeframes, these systems enable data-driven formulation optimization, warranty validation, and regulatory compliance. The integration of precise UV spectral control, moisture cycling, and temperature management creates realistic weathering conditions that predict long-term coating performance across diverse applications and climates.
FAQsHow long does accelerated UV testing take compared to outdoor exposure?
Accelerated testing typically compresses 1-3 years of outdoor weathering into 1000-2000 hours of chamber exposure, depending on the coating type and testing protocol. Acceleration factors between 4:1 and 12:1 are common, though correlation studies with outdoor exposure data are essential for accurate lifetime predictions.
Can UV resistance chambers test coatings on actual product components?
Modern chambers accommodate three-dimensional specimens, enabling testing of coated parts rather than just flat panels. The UV-SI-260 features removable sample holders designed for both flat and complex geometries, allowing manufacturers to evaluate production parts under controlled weathering conditions that replicate end-use performance.
What wavelength setting should be used for coating evaluation?
UVA-340 lamps at 340nm best simulate natural sunlight's long-term degradation effects for most coating applications. UVB-313 lamps at 313nm provide faster, more aggressive testing useful for comparative screening. Selection depends on whether realistic outdoor correlation or accelerated comparative evaluation is the primary objective.
Contact LIB Industry - your trusted UV resistance test chamber manufacturer and supplier - to discuss customized weathering evaluation solutions for your coating development programs. Our technical team provides comprehensive support from equipment selection through protocol development and results interpretation. Reach us at ellen@lib-industry.com to enhance your coating testing capabilities.
Aging Studies of Processed Food Using Hot and Cold Chambers
Aging studies of processed food using hot and cold chambers enable manufacturers to simulate extended storage conditions in compressed timeframes, evaluating how temperature fluctuations and humidity levels affect product stability, packaging integrity, and sensory characteristics. These controlled environmental chambers replicate real-world storage scenarios - from warehouse conditions to transportation stress - allowing food scientists to predict shelf life accurately, optimize formulations, and ensure regulatory compliance before products reach consumers. By accelerating natural aging processes through precise climate control, manufacturers gain critical insights into degradation patterns, microbial growth potential, and quality retention that inform packaging decisions and expiration dating.
Why Aging Studies Matter for Processed Food?Consumer Safety and Regulatory Compliance
Food manufacturers face stringent regulatory requirements regarding shelf-life claims and expiration dating. Aging studies provide the empirical evidence needed to substantiate these claims, demonstrating that products maintain safety and quality throughout their intended lifespan. Regulatory bodies worldwide require comprehensive stability data before approving shelf-life designations, making these studies essential for market entry and continued compliance.
evidence needed to substantiate these claims, demonstrating that products maintain safety and quality throughout their intended lifespan. Regulatory bodies worldwide require comprehensive stability data before approving shelf-life designations, making these studies essential for market entry and continued compliance.
Economic Impact of Shelf Life Optimization
Extending product shelf life by even a few days can significantly reduce waste and improve distribution efficiency. Aging studies help identify the optimal balance between formulation costs and product longevity, enabling manufacturers to make data-driven decisions about preservatives, packaging materials, and storage recommendations that maximize profitability while maintaining quality standards.
Brand Reputation and Quality Assurance
Premature product deterioration damages brand credibility and consumer trust. Through systematic aging studies, manufacturers can identify potential failure points before widespread distribution, ensuring that products consistently meet quality expectations throughout their shelf life, thereby protecting brand reputation and reducing costly recalls.
Effects of Temperature and Humidity on Food QualityChemical Degradation Pathways
Temperature fluctuations accelerate oxidative reactions, enzymatic browning, and lipid rancidity in processed foods. Hot and cold chambers allow researchers to quantify reaction rates at various temperatures, applying Arrhenius kinetics to predict degradation under ambient conditions. Humidity similarly affects moisture-sensitive reactions, including non-enzymatic browning and texture changes in low-moisture products.
Physical Changes and Structural Integrity
Thermal cycling induces physical transformations such as fat bloom in chocolate, moisture migration in layered products, and crystallization in confections. Controlled humidity exposure affects crispness in crackers, caking in powdered products, and syneresis in gel-based foods. These chambers enable systematic documentation of physical deterioration patterns.
Microbial Growth Correlation
Temperature and humidity directly influence microbial proliferation rates. Chambers maintaining specific conditions allow researchers to establish growth curves for spoilage organisms and pathogens, identifying critical control points for preservation strategies and validating antimicrobial interventions under realistic storage scenarios.
|
Environmental Factor |
Primary Impact |
Affected Product Categories |
|
High Temperature (>30°C) |
Accelerated oxidation, vitamin degradation |
Oils, fortified products, snacks |
|
Low Temperature (<5°C) |
Retrogradation, texture hardening |
Bakery products, sauces |
|
High Humidity (>70% RH) |
Moisture absorption, microbial growth |
Dried fruits, cereals, powders |
|
Low Humidity (<30% RH) |
Moisture loss, texture changes |
Fresh-cut products, refrigerated items |
Accelerated Shelf Life Testing Techniques

Temperature Cycling Protocols
Accelerated aging employs elevated temperatures to compress months of shelf life into weeks of testing. The Q10 approach - where reaction rates double with every 10°C increase - provides mathematical frameworks for extrapolating accelerated data to ambient conditions. Hot and cold chambers enable precise temperature cycling that mimics seasonal variations and distribution chain realities.
Humidity Stress Testing
Controlled humidity challenges reveal moisture barrier effectiveness in packaging and product hygiene sensitivity. By exposing samples to extreme humidity levels (20-98% RH range), researchers identify moisture-related failure modes and validate packaging specifications, ensuring products withstand humid climates and refrigerated storage condensation.
Combined Environmental Stressors
Real-world conditions involve simultaneous temperature and humidity variations. Advanced testing protocols incorporate cyclic conditions - alternating between hot/humid and cool/dry phases - to simulate distribution through varied climate zones, revealing synergistic effects that single-factor testing might miss.
Monitoring Packaging and Product IntegrityBarrier Property Evaluation
Packaging materials exhibit temperature-dependent permeability to oxygen, moisture, and volatile compounds. Hot and cold chambers allow systematic evaluation of barrier degradation over time, ensuring packaging maintains protective functions throughout the product's shelf life. Seal integrity testing under thermal stress identifies potential failure points before distribution.
Headspace Gas Analysis
Modified atmosphere packaging relies on maintaining specific gas compositions. Chamber studies track headspace oxygen depletion and carbon dioxide evolution, validating respiration models and ensuring gas barrier properties remain effective under various temperature conditions, preventing premature spoilage.
Visual and Structural Defects
Thermal expansion and contraction stress packaging materials, potentially causing delamination, seal failures, or label adhesion problems. Systematic chamber exposure reveals these defects, enabling packaging optimization before commercial production begins.
|
Testing Parameter |
Measurement Frequency |
Acceptance Criteria |
|
Oxygen transmission rate |
Weekly |
<1% increase from baseline |
|
Seal strength |
Bi-weekly |
>80% initial strength retained |
|
Moisture content |
Weekly |
Within ±5% of target |
|
Package appearance |
Daily |
No visible defects |
Sensory and Microbial Assessment of Aged FoodOrganoleptic Property Tracking
Trained sensory panels evaluate color, aroma, flavor, and texture attributes at predetermined intervals throughout chamber studies. Quantitative descriptive analysis identifies specific attribute changes, establishing sensory shelf-life endpoints that often precede safety concerns but determine consumer acceptance.
Microbial Enumeration Protocols
Regular microbiological testing throughout aging studies tracks total plate counts, yeast and mold levels, and specific pathogen indicators. Hot and cold chambers maintaining specific conditions enable validation of microbial growth models, ensuring products remain below regulatory limits throughout their shelf life.
Correlating Sensory and Microbial Data
Integrating sensory and microbiological data reveals whether sensory degradation or microbial growth limits shelf life. This correlation guides formulation adjustments - whether improving preservative systems or enhancing flavor stability - to extend the limiting factor.
Predictive Analysis for Product Shelf Life
| Name | shelf life test chamber | |||||
|
Model |
TH-100 |
|||||
|
Temperature range |
-20℃ ~+150 ℃ |
|||||
|
Low type |
A: -40℃ B:-70℃ C -86℃ |
|||||
|
Humidity Range |
20%-98%RH |
|||||
|
Temperature deviation |
± 2.0 ℃ |
|||||
|
Heating rate |
3 ℃ / min |
|||||
|
Cooling rate |
1 ℃ / min |
|||||
|
Controller |
Programmable color LCD touch screen controller, Multi-language interface, Ethernet , USB |
|||||
|
Exterior material |
Steel Plate with protective coating |
|||||
|
Interior material |
SUS304 stainless steel |
|||||
|
Standard configuration |
1 Cable hole (Φ 50) with plug; 2 shelves |
|||||
|
Timing Function |
0.1~999.9 (S,M,H) settable |
|||||
Mathematical Modeling Approaches
Kinetic models transform accelerated aging data into shelf-life predictions at distribution temperatures. Zero-order, first-order, and Arrhenius equations describe degradation rates, with statistical validation ensuring prediction accuracy. Hot and cold chambers-generated data provides the quantitative foundation for these models.
Statistical Confidence Intervals
Shelf-life determinations incorporate statistical rigor, establishing confidence intervals that account for batch variability and measurement uncertainty. Replicated chamber studies across multiple production lots ensure predictions reflect real-world variability, not isolated batch performance.
Real-Time Validation Studies
Accelerated predictions require validation through real-time storage studies at ambient conditions. Parallel chamber testing and warehouse storage confirm model accuracy, building regulatory confidence and refining prediction algorithms for future products.
|
Model Type |
Application |
Data Requirements |
Prediction Accuracy |
|
Arrhenius |
Temperature-dependent reactions |
3+ temperature points |
±15-20% typical |
|
Weibull |
Microbial shelf life |
Time-series counts |
±10-15% typical |
|
Linear regression |
Sensory degradation |
Multiple timepoints |
±20-25% typical |
Accelerate Food Shelf-Life Studies with LIB Industry's Reliable Hot and Cold ChamberAdvanced Temperature and Humidity Control
LIB Industry hot and cold chambers deliver precise environmental control across -70°C to +150°C temperature ranges and 20-98% RH humidity ranges. Programmable LCD touch screen controllers enable complex cycling protocols, while automatic water supply systems maintain consistent humidity without manual intervention, ensuring uninterrupted long-term studies.

Scalable Chamber Configurations
From benchtop 100L models to walk-in 1000L chambers, LIB Industry offers solutions matching diverse testing requirements. Multiple sample shelves and cable ports accommodate simultaneous testing of numerous products and integrated monitoring equipment, maximizing research efficiency and data generation.
Comprehensive Safety and Monitoring Features
Built-in protection systems - including over-temperature, refrigerant high-pressure, and earth leakage safeguards - ensure sample integrity and operator safety. Ethernet connectivity enables remote monitoring and data logging, creating comprehensive documentation for regulatory submissions and quality management systems.
Conclusion
Hot and cold chambers transform food shelf-life determination from prolonged guesswork into systematic science. Through controlled environmental challenges, manufacturers gain predictive insights into product behavior, optimize formulations and packaging, and confidently establish expiration dates backed by empirical evidence. These capabilities reduce development timelines, minimize waste, and ensure consistent quality delivery.
FAQs
How long does accelerated aging testing typically take compared to real-time studies?
Accelerated testing at elevated temperatures can compress 12-24 months of ambient shelf life into 3-6 months of chamber testing, depending on the temperature differential and product characteristics. Validation with real-time studies confirms accuracy.
Can hot and cold chambers simulate transportation stress on packaged foods?
Absolutely. Programmable cycling between temperature extremes replicates truck and container transport conditions, revealing packaging vulnerabilities and product responses to distribution chain thermal stress that static storage testing cannot identify.
What sample size is needed for statistically valid shelf-life studies?
Minimum three production batches with triplicate samples per timepoint ensures statistical validity. Larger sample sizes improve confidence intervals, particularly when establishing shelf-life endpoints for regulatory submissions requiring high certainty levels.
Partner with LIB Industry, a leading hot and cold chamber manufacturer and supplier, for comprehensive environmental testing solutions. Our turn-key services include chamber design, installation, training, and ongoing support. Contact ellen@lib-industry.com to discuss your food aging study requirements and accelerate your product development.
Using a Shelf Life Test Chamber for ICH Stability Compliance
A year after acquiring our product, Rob, a procurement manager at an electronics company, carried out a Shelf Life Test Chamber. He expressed his satisfaction, noting, 'Even though the chamber hasn't been used much recently, I'm confident it's still working effectively.' Now, let's explore the Shelf Life Test Chamber in detail.
Meeting ICH stability compliance requires precise environmental control to evaluate how pharmaceutical products, medical devices, and nutraceuticals maintain their quality over time. A shelf life test chamber provides the controlled temperature and humidity conditions mandated by ICH guidelines, enabling manufacturers to generate reliable stability data for regulatory submissions. These specialized chambers replicate long-term storage scenarios and accelerated aging conditions, helping companies demonstrate product safety and efficacy throughout the intended shelf life while satisfying stringent requirements from regulatory authorities worldwide.
What are ICH Guidelines for Stability Testing?The Harmonization Framework
The International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) established unified stability testing protocols to streamline regulatory submissions across different regions. These guidelines eliminate redundant testing by creating standardized methodologies accepted by regulatory bodies in Europe, Japan, the United States, and other participating countries. The framework addresses drug substances, finished pharmaceutical products, and biotechnological materials through comprehensive quality assessment protocols.
Core ICH Stability Protocols
ICH Q1A(R2) defines the fundamental stability testing requirements, specifying storage conditions, testing frequencies, and minimum data requirements. The guideline mandates evaluation under defined temperature and humidity combinations that represent different climatic zones. Zone I encompasses temperate climates, Zone II covers subtropical regions, Zone III represents hot and dry conditions, while Zone IV addresses hot and humid environments. Manufacturers must select appropriate testing conditions based on their intended markets.
Regulatory Acceptance Criteria
Stability studies following ICH guidelines must demonstrate that active pharmaceutical ingredients maintain specified potency levels, physical characteristics remain acceptable, and degradation products stay within established limits. The data generated forms the scientific basis for determining expiration dates, establishing storage recommendations, and supporting shelf life claims on product labels. Regulatory agencies evaluate this information during new drug applications, abbreviated new drug applications, and variation submissions.
Key Parameters in ICH-Compliant TestingTemperature Control Requirements
Precise temperature maintenance represents a critical element of ICH-compliant stability testing. Long-term studies typically require 25°C ± 2°C, while accelerated testing demands 40°C ± 2°C. The shelf life test chamber must maintain these setpoints consistently throughout extended study periods, with minimal deviation. Advanced refrigeration systems using environmentally friendly refrigerants like R404A and R23 ensure reliable performance across the required temperature spectrum from -86°C to +150°C.
while accelerated testing demands 40°C ± 2°C. The shelf life test chamber must maintain these setpoints consistently throughout extended study periods, with minimal deviation. Advanced refrigeration systems using environmentally friendly refrigerants like R404A and R23 ensure reliable performance across the required temperature spectrum from -86°C to +150°C.
Humidity Management Systems
Relative humidity control complements temperature regulation in creating accurate stability conditions. ICH guidelines specify 60% RH ± 5% for long-term testing and 75% RH ± 5% for accelerated studies. Shelf life test chambers employ forced air convection systems with dry and wet bulb humidity sensors to achieve uniform moisture distribution. The 20%-98% RH range available in modern equipment accommodates various testing scenarios, including intermediate conditions and stress testing protocols.
Uniformity and Distribution
Spatial uniformity throughout the testing chamber ensures all samples experience identical environmental exposure. Temperature deviation should not exceed ± 2.0°C between different locations within the workspace. Mirror-finish stainless steel interiors with optimized air circulation patterns minimize hot spots and cold zones. Multiple sample shelves with perforated designs facilitate airflow while accommodating substantial product quantities during simultaneous testing campaigns.
|
Parameter |
Long-Term Condition |
Accelerated Condition |
Intermediate Condition |
|
Temperature |
25°C ± 2°C |
40°C ± 2°C |
30°C ± 2°C |
|
Relative Humidity |
60% ± 5% RH |
75% ± 5% RH |
65% ± 5% RH |
|
Minimum Duration |
12 months |
6 months |
6 months |
Designing Test Protocols for Regulatory ApprovalSample Selection and Placement
Protocol development begins with representative sample selection from commercial production batches manufactured using validated processes. At minimum, three batches should undergo stability evaluation to demonstrate consistency. Sample placement within the chamber must account for potential microenvironment variations, with positioning documented in the protocol. Rotation schedules may be implemented to eliminate bias from location-specific effects.
Testing Timepoint Determination
ICH guidelines specify minimum testing frequencies for stability programs. Long-term studies require evaluation at 0, 3, 6, 9, 12, 18, and 24 months, with annual testing continuing thereafter. Accelerated studies demand assessment at 0, 1, 2, 3, and 6 months. Intermediate conditions follow a 0, 6, 9, and 12-month schedule. The protocol should define exact timepoint windows and procedures for handling scheduling deviations.
Analytical Method Validation
All analytical procedures used for stability assessment require validation demonstrating specificity, linearity, accuracy, precision, and stability-indicating capability. Methods must detect and quantify degradation products formed under stress conditions. The protocol should reference validated method documents and specify acceptance criteria aligned with product specifications and regulatory commitments. Trending analysis procedures help identify subtle changes preceding specification failures.
Long-term vs Accelerated Stability TestingLong-term Study Characteristics
Long-term stability testing evaluates products under recommended storage conditions over extended periods matching or exceeding the proposed shelf life. This approach generates real-time data reflecting actual stability performance, providing the most reliable foundation for expiration dating. Studies continue at least 12 months beyond the proposed shelf life to demonstrate adequate stability margins. The data supports primary stability claims submitted to regulatory authorities.
| Name | shelf life test chamber | |||||
|
Model |
TH-100 |
|||||
|
Temperature range |
-20℃ ~+150 ℃ |
|||||
|
Low type |
A: -40℃ B:-70℃ C -86℃ |
|||||
|
Humidity Range |
20%-98%RH |
|||||
|
Temperature deviation |
± 2.0 ℃ |
|||||
|
Heating rate |
3 ℃ / min |
|||||
|
Cooling rate |
1 ℃ / min |
|||||
|
Controller |
Programmable color LCD touch screen controller, Multi-language interface, Ethernet , USB |
|||||
|
Exterior material |
Steel Plate with protective coating |
|||||
|
Interior material |
SUS304 stainless steel |
|||||
|
Standard configuration |
1 Cable hole (Φ 50) with plug; 2 shelves |
|||||
|
Timing Function |
0.1~999.9 (S,M,H) settable |
|||||
Accelerated Testing Applications
Accelerated conditions use elevated temperature and humidity to induce faster degradation, predicting long-term stability behavior within compressed timeframes. The Arrhenius relationship between temperature and reaction rate underpins this extrapolation. Significant changes observed during accelerated testing trigger additional intermediate condition studies or necessitate protective packaging modifications. Accelerated data supports preliminary shelf life estimates during product development and provides early warning of potential stability issues.
Combining Study Types
Comprehensive stability programs incorporate both long-term and accelerated testing in complementary roles, typically conducted within a shelf life test chamber to ensure controlled and repeatable environmental conditions. Accelerated studies enable rapid formulation screening and process optimization during development phases. Long-term testing subsequently confirms predicted stability and establishes definitive shelf life claims. The combination provides both timely development insights and robust regulatory data. Ongoing commitment batches enter long-term programs to monitor commercial production stability throughout the product lifecycle.
|
Study Type |
Primary Purpose |
Temperature/RH |
Duration |
Data Application |
|
Long-term |
Real-time stability |
25°C/60% RH |
12-36+ months |
Primary shelf life claim |
|
Accelerated |
Predictive assessment |
40°C/75% RH |
6 months |
Supporting data, early warning |
|
Intermediate |
Confirmation studies |
30°C/65% RH |
12 months |
Additional support when accelerated fails |
Documentation and Reporting for ICH ComplianceProtocol Documentation Requirements
Comprehensive protocols form the foundation of compliant stability programs. Documents must clearly state the study objective, product identification, batch information, storage conditions, sampling schedule, analytical methods, acceptance criteria, and statistical evaluation plans. Deviation procedures, sample tracking systems, and change control processes require detailed description. Regulatory reviewers assess protocol adequacy during application evaluation, making thoroughness essential.
Data Recording and Traceability
Modern programmable controllers with color LCD touchscreens enable automated data logging of chamber conditions throughout study duration. Ethernet connectivity facilitates remote monitoring and data download via USB interfaces. Complete environmental records demonstrate continuous compliance with specified conditions. Any excursions require documentation with impact assessments determining whether affected samples remain suitable for evaluation or require replacement.
Regulatory Submission Formats
Stability reports summarize study results in formats specified by regional regulatory guidance. The Common Technical Document (CTD) format organizes stability data within Module 3 for pharmaceutical applications. Reports should include batch analyses, tabulated results with statistical trending, graphical presentations of key parameters, and scientific interpretation of findings. Conclusions must clearly support the proposed shelf life and storage conditions.
Benefits of Test Chambers in Meeting ICH StandardsPrecision and Reproducibility
Dedicated stability chambers eliminate environmental variability that compromises study integrity. Heating rates of 3°C/min and cooling rates of 1°C/min enable rapid condition transitions between different study phases. Temperature fluctuation within ± 0.5°C ensures setpoint stability during extended operation. This precision generates reproducible data suitable for regulatory decision-making and supports comparability assessments across different study timepoints.

Multi-zone Testing Capability
Advanced shelf life test chambers accommodate simultaneous studies at different ICH conditions, maximizing laboratory efficiency. Independent compartments maintain distinct temperature and humidity combinations, enabling parallel evaluation of long-term, accelerated, and intermediate scenarios. This capability proves particularly valuable during product development when multiple formulations undergo comparative stability assessment. Consolidated testing reduces equipment footprint and operational costs.
Enhanced Safety Features
Equipment design incorporating separated water and electrical systems minimizes safety risks during continuous long-term operation. Mirror-finish stainless steel interiors resist corrosion from humidity exposure and facilitate cleaning when product spillage or contamination occurs. Customization options include explosion-proof configurations for testing volatile formulations and low-pressure chambers simulating high-altitude storage conditions. These features expand testing capabilities while maintaining personnel safety.
|
Chamber Feature |
ICH Compliance Benefit |
Operational Advantage |
|
Programmable controller |
Automated condition cycling |
Reduced manual intervention |
|
Network connectivity |
Real-time monitoring |
Remote oversight capability |
|
Uniform air circulation |
Consistent sample exposure |
Eliminates position effects |
|
Dual refrigeration system |
Wide temperature range |
Accommodates stress testing |
Simplify ICH Compliance with LIB Industry's Validated Shelf Life Test ChamberProven Performance Specifications
The TH-225 and TH-500 models from Xi'an LIB Environmental Simulation Industry deliver the temperature precision and humidity control demanded by ICH protocols. With internal dimensions accommodating substantial sample quantities (500×600×750mm and 700×800×900mm respectively), these chambers support comprehensive stability programs. The -86°C to +150°C temperature range exceeds ICH requirements, enabling stress testing and specialized applications beyond standard stability conditions.
demanded by ICH protocols. With internal dimensions accommodating substantial sample quantities (500×600×750mm and 700×800×900mm respectively), these chambers support comprehensive stability programs. The -86°C to +150°C temperature range exceeds ICH requirements, enabling stress testing and specialized applications beyond standard stability conditions.
Quality Manufacturing Standards
CE-approved construction demonstrates compliance with international safety and performance standards. High-precision PT100Ω/MV A-class temperature probes provide accurate sensing, while forced air convection ensures uniform distribution throughout the workspace. Environmentally friendly refrigerants align with global sustainability initiatives without compromising performance. Punch-design sample shelves fabricated from #304 mirror-finish stainless steel combine durability with easy maintenance.
Customization and Support
LIB Industry's customization capabilities address unique testing requirements beyond standard configurations. Explosion-proof modifications accommodate flammable materials, low-pressure options simulate transportation or high-altitude conditions, and vibration integration enables combined environmental stress testing. The company provides complete turn-key solutions encompassing research, design, production, commissioning, delivery, installation, and operator training. This comprehensive approach ensures seamless integration of stability testing capabilities into pharmaceutical quality systems.
Conclusion
Achieving ICH stability compliance demands environmental test equipment capable of maintaining precise temperature and humidity conditions throughout extended study periods. Shelf life test chambers provide the controlled environments necessary for generating reliable stability data supporting regulatory submissions worldwide. By combining accurate parameter control, comprehensive documentation capabilities, and validated performance, these specialized chambers enable pharmaceutical manufacturers to demonstrate product quality and establish defensible shelf life claims that satisfy regulatory requirements while protecting patient safety.
FAQsWhat temperature tolerance is acceptable for ICH stability studies?
ICH guidelines permit ± 2°C deviation from the specified storage temperature during long-term and accelerated stability testing. Modern chambers typically achieve tighter control within ± 0.5°C fluctuation, providing additional assurance that samples remain within acceptable environmental ranges throughout the study duration.
How many batches require stability testing for regulatory submissions?
Regulatory authorities generally require stability data from at least three commercial-scale batches manufactured using validated production processes. These batches should represent the proposed commercial formulation and packaging configuration, demonstrating consistency of stability performance across multiple manufacturing campaigns.
Can accelerated stability data replace long-term studies?
Accelerated testing provides supporting evidence but cannot substitute for long-term stability data when establishing primary shelf life claims. Regulatory agencies require real-time stability information at recommended storage conditions for final approval, though accelerated studies help predict behavior and support interim shelf life assignments during product development.
Partner with LIB Industry for Your Stability Testing Needs
As a leading manufacturer and supplier of environmental test chambers, LIB Industry delivers validated shelf life testing solutions that streamline ICH compliance. Our factory produces quality equipment at competitive prices, backed by comprehensive technical support. Contact ellen@lib-industry.com to discuss your stability testing requirements and request a quotation.
Dust Proof Test Chamber Applications in EV Electrical System Testing
Electric vehicles depend on sophisticated electrical architectures that must withstand challenging environmental conditions throughout their operational lifespan. Dust intrusion poses significant threats to battery management systems, inverters, charging ports, and control modules. A dust proof test chamber enables manufacturers to simulate real-world particulate exposure scenarios, validating whether protective housings maintain IP ratings under extreme conditions. By subjecting high-voltage components to controlled dust concentrations (2-4kg/m³), engineers identify potential vulnerabilities in sealing mechanisms, gasket performance, and connector interfaces before vehicles reach consumers. This proactive validation approach reduces warranty claims, enhances safety certifications, and ensures long-term reliability in diverse operating environments.
Why EV Systems Require Dust Resistance Validation?Complex Electrical Architectures Demand Protection
Modern electric vehicles integrate hundreds of interconnected electrical components operating at voltages exceeding 800V. These systems include battery packs with thousands of individual cells, power distribution units managing kilowatt-level loads, and thermal management circuits maintaining optimal operating temperatures. Each connection point, junction box, and cable gland represents a potential entry path for airborne particulates.
battery packs with thousands of individual cells, power distribution units managing kilowatt-level loads, and thermal management circuits maintaining optimal operating temperatures. Each connection point, junction box, and cable gland represents a potential entry path for airborne particulates.
Environmental Exposure During Vehicle Operation
Electric vehicles encounter varied dust exposure throughout their service life. Urban environments generate brake dust particles, tire wear debris, and construction site particulates. Rural and off-road applications expose underbody components to agricultural dust, sand, and soil particles. Coastal regions introduce salt-laden aerosols that accelerate corrosion when combined with particulate infiltration.
Regulatory Compliance Requirements
Automotive standards including ISO 20653 and IEC 60529 establish specific ingress protection criteria for electrical enclosures. Achieving IP6X certification requires demonstrating complete dust-tightness under vacuum conditions with talcum powder at specified concentrations. Verification testing in controlled chamber environments provides documented evidence supporting certification claims and regulatory approvals.
Dust Exposure Scenarios for Electric Vehicle ComponentsBattery Pack Enclosure Validation
Battery management systems monitor individual cell voltages, temperatures, and balancing circuits through complex wiring harnesses. Dust infiltration into battery enclosures can create conductive paths between adjacent terminals, increasing self-discharge rates and creating short-circuit risks. Testing chambers apply calibrated talcum powder (50μm wire diameter standards) to verify that gasket compression, vent filter performance, and weld seam integrity prevent particulate ingress.
Motor Controller and Inverter Testing
Power electronics converting DC battery voltage to three-phase AC motor drive signals generate significant heat requiring active cooling systems. Air intake filters protecting these thermal management pathways require validation under dust loading conditions. Dust proof test chamber testing evaluates filter efficiency degradation, airflow restriction accumulation, and particulate bypass through gasket interfaces.
Charging System Component Evaluation
Onboard chargers, charge port assemblies, and cable connectors experience repeated exposure during vehicle operation and charging events. Testing protocols simulate dust accumulation on electrical contacts, assess insulation resistance degradation, and verify that protective covers maintain sealing effectiveness through multiple operation cycles.
|
Component Category |
Primary Dust Vulnerability |
Test Focus Area |
|
Battery Pack Housing |
Gasket interfaces, vent filters |
Complete enclosure sealing |
|
Power Electronics |
Cooling air intakes, connector seals |
Thermal management integrity |
|
Charging Systems |
Contact surfaces, cable interfaces |
Repeated operation durability |
|
Sensor Assemblies |
Optical surfaces, signal connectors |
Measurement accuracy retention |
Testing High-Voltage Modules under Dust ConditionsControlled Environment Simulation
Chambers equipped with programmable temperature control (ambient to +50℃) replicate thermal cycling conditions that affect gasket compression and housing expansion. Adjustable vacuum systems create pressure differentials simulating altitude changes and thermal breathing effects. Dust circulation mechanisms maintain uniform particulate distribution throughout the test volume, ensuring consistent exposure across all component surfaces.
|
|
|
| Name | dust proof test chamber |
|
Workroom dimension (mm) |
800*800*800 D*W*H |
|
External dimension (mm) |
950*3150*1800 D*W*H |
|
Interior Volume (L) |
510 |
|
Diameter of Turntable (mm) |
600 |
|
Turntable loads |
20kgs Max |
|
Turntable Rotation Speed |
0~7r/min (Adjustable) |
|
Internal Diameter of IPX5 Nozzle |
6.3 mm |
|
Internal Diameter of IPX6 Nozzle |
12.5 mm |
|
Water Flow Rate IPX5/ IPX6 |
12.5L/min ±5% / 100L/min ±5% |
|
Controller |
Programmable color LCD touch screen controller |
|
Ethernet connection, PC Link, USB |
|
|
Build-in Water Tank(mm) |
370*375*950 |
|
View Window Size(mm) |
475*475 |
Standardized Test Protocols
Validation procedures specify exposure duration, particle concentration, and environmental conditions matching anticipated service environments. Typical protocols include 8-hour exposure cycles at 2kg/m³ concentration, followed by disassembly inspections documenting particulate accumulation locations. Electrical performance verification measures insulation resistance, dielectric strength, and contact resistance changes attributable to dust exposure.
Performance Metric Documentation
Testing generates quantitative data supporting design validation decisions. Insulation resistance measurements exceeding 100MΩ confirm adequate sealing performance. Contact resistance stability within specified tolerances validates connector sealing effectiveness. Thermal imaging identifies localized heating caused by particulate accumulation on heat transfer surfaces.
Sealing and Insulation Performance EvaluationGasket Compression Analysis
Elastomeric seals maintaining dust-tight enclosures experience compression set degradation under sustained loading and temperature exposure. Dust proof test chamber testing combined with accelerated aging protocols evaluates long-term sealing effectiveness. Post-exposure compression force measurements quantify gasket recovery characteristics and identify potential seal failure modes.
Dust proof test chamber testing combined with accelerated aging protocols evaluates long-term sealing effectiveness. Post-exposure compression force measurements quantify gasket recovery characteristics and identify potential seal failure modes.
Interface Integrity Assessment
Multi-part housings rely on precise manufacturing tolerances maintaining gap dimensions below particulate sizes. Testing reveals whether assembly variations, thermal expansion mismatches, or vibration-induced settling compromises sealing interfaces. Disassembly inspections document dust accumulation patterns indicating preferential infiltration pathways requiring design modifications.
Electrical Isolation Verification
High-voltage systems maintain safety through multiple isolation barriers between energized conductors and chassis ground. Dust accumulation on insulating surfaces can reduce breakdown voltages and create tracking paths under moisture exposure. Testing protocols measure insulation resistance at elevated voltages (typically 2x operating voltage plus 1000V) confirming adequate safety margins post-exposure.
|
Test Parameter |
Acceptance Criteria |
Measurement Method |
|
Insulation Resistance |
>100MΩ at 500VDC |
Megohmmeter testing |
|
Contact Resistance |
<5mΩ increase |
Four-wire measurement |
|
Enclosure Integrity |
Zero particulate ingress |
Visual inspection after disassembly |
Accelerated Dust Aging for EV Electrical SystemsCompressed Timeline Validation
Development schedules require validating 10-15 year component lifespans within months. Accelerated testing protocols combine elevated dust concentrations, increased cycle frequencies, and temperature extremes compressing equivalent operational exposures. Chamber programmability enables automated test sequences replicating years of environmental cycling.
Correlation to Field Performance
Establishing acceleration factors requires correlating dust proof test chamber test results with field failure data from operating vehicle fleets. Statistical analysis comparing exposure conditions, failure modes, and component degradation rates validates that chamber testing accurately predicts real-world performance. This correlation enables confident extrapolation from short-duration tests to long-term reliability projections.
Failure Mode Identification
Accelerated testing reveals potential failure mechanisms before mass production deployment. Common dust-related failures include connector fretting corrosion, gasket degradation allowing moisture ingress, and particulate accumulation blocking thermal management pathways. Early identification enables design modifications preventing costly field failures and recall campaigns.
Ensuring Safety and Reliability in EV OperationFire Risk Mitigation
Dust accumulation on high-current terminals can create resistive heating leading to thermal runaway conditions. Validation testing confirms that protective housings prevent dangerous particulate accumulation. Thermal imaging during energized testing identifies potential hot spots requiring design attention.
Electromagnetic Compatibility Protection
Dust infiltration into shielded enclosures compromises electromagnetic interference containment. Testing verifies that sealing maintains RF shielding effectiveness preventing interference with communication systems, sensor networks, and adjacent electronic modules. Conducted and radiated emissions testing post-dust exposure quantifies shielding degradation.
Long-term Warranty Support
Comprehensive dust resistance validation reduces warranty exposure by identifying potential failure modes before customer delivery. Documented test results support warranty claims analysis, distinguishing manufacturing defects from environmental damage. This documentation protects manufacturers from unwarranted liability while ensuring legitimate failures receive appropriate coverage.
|
Reliability Aspect |
Dust Chamber Validation Benefit |
Impact on Vehicle Safety |
|
Thermal Management |
Confirms cooling path integrity |
Prevents component overheating |
|
Electrical Isolation |
Validates insulation performance |
Eliminates shock hazards |
|
Connector Durability |
Verifies contact protection |
Maintains signal integrity |
Power EV Durability with LIB Industry's High-Performance Dust Proof Test ChamberAdvanced Testing Capabilities
LIB Industry's dust proof test chambers provide precise environmental control supporting comprehensive validation programs. Programmable color LCD touchscreen controllers enable complex test sequence programming. Ethernet connectivity facilitates remote monitoring and data logging supporting quality management system integration. Internal volumes ranging from 800L to 1000L accommodate complete battery modules, inverter assemblies, and full charging system components.

Compliance-Ready Testing Platform
Equipment designed to IEC 60529 and ISO 20653 standards ensures test results satisfy regulatory requirements. Calibrated dust circulation systems maintain uniform concentrations throughout the exposure period. Integrated vacuum systems with precision pressure regulation replicate standardized test conditions. Documentation packages support certification submissions and audit requirements.
Manufacturing Support Services
Beyond equipment supply, LIB Industry provides comprehensive technical support including test protocol development, operator training, and ongoing calibration services. Applications engineering assistance helps customers optimize test procedures for specific component geometries and validation requirements. This turn-key solution approach accelerates time-to-market while ensuring robust validation data quality.
Conclusion
Dust resistance validation represents a critical quality gate ensuring electric vehicle electrical systems withstand real-world environmental challenges. Comprehensive testing in controlled chamber environments identifies design vulnerabilities, validates protective measures, and generates documentation supporting regulatory compliance. As EV adoption accelerates globally, manufacturers requiring reliable dust proof test chambers gain competitive advantages through reduced warranty costs, enhanced safety records, and accelerated development cycles.
FAQs
What dust concentration levels do automotive standards require for EV component testing?
ISO 20653 and IEC 60529 specify talcum powder at 2kg/m³ concentration for IP6X certification testing. The standardized test uses particles with 50μm nominal wire diameter and 75μm nominal gap width between wires, creating representative worst-case exposure conditions for electrical enclosures.
How long should dust exposure testing run for reliable EV component validation?
Standard validation protocols typically specify 8-hour continuous exposure followed by component inspection and electrical performance verification. Accelerated aging programs may extend exposure to 99+ hours at elevated temperatures and dust concentrations, simulating years of operational exposure within compressed timeframes.
Can dust proof test chambers accommodate complete battery pack assemblies?
Modern chambers with 1000L internal volumes easily accommodate full battery modules and large inverter housings. The 1000x1000x1000mm internal dimensions provide sufficient clearance for mounting fixtures while maintaining uniform dust distribution across all component surfaces during testing procedures.
Contact LIB Industry for Your Testing Solutions
LIB Industry, a leading environmental test chamber manufacturer and supplier, delivers high-quality dust proof test chambers backed by comprehensive technical support. Our factory produces CE-certified equipment meeting international standards for automotive component validation. Contact our team at ellen@lib-industry.com to discuss your specific testing requirements and receive a detailed quotation.
UV Resistance Test Chamber Applications in Automotive Plastic Parts Testing
Several months after installing the Light Resistance Test Chamber, an experienced instrumentation professional from India shared his feedback:“We have installed and serviced weathering instruments for many years, including systems from leading European brands. After using the LIB Light Resistance Test Chamber, we found its performance and test reliability to be on par with ATLAS, while offering a more competitive price. The instrument quality fully meets our expectations, and we are very satisfied with its overall performance.”This feedback highlights the chamber's proven stability, consistent light exposure control, and strong value for long-term material testing applications.
Automotive plastic parts face relentless exposure to ultraviolet radiation throughout their service life, making UV resistance testing essential for quality assurance. A UV resistance test chamber enables manufacturers to simulate years of sunlight exposure within weeks, predicting how dashboard components, bumpers, exterior trim, and interior plastics will perform under real-world conditions. This accelerated weathering approach identifies potential failures - such as discoloration, cracking, or mechanical degradation - before parts reach production lines. By subjecting materials to controlled UV radiation, temperature cycles, and moisture conditions, automotive engineers can optimize material formulations, validate protective coatings, and ensure components meet stringent durability standards demanded by modern vehicle lifespans.
Why UV Resistance is Critical for Automotive Plastics?
Material Degradation Mechanisms
Ultraviolet radiation triggers photochemical reactions within polymer chains, breaking molecular bonds and initiating oxidative degradation. This photodegradation process weakens the structural integrity of automotive plastics, reducing their tensile strength and impact resistance over time. Polypropylene, ABS, polycarbonate, and other common automotive polymers exhibit varying susceptibility to UV-induced damage, necessitating material-specific testing protocols.
Economic Implications of UV Damage
Premature failure of plastic components generates substantial warranty costs and damages brand reputation. Dashboard cracking, faded exterior trim, and brittle weather stripping represent common UV-related failures that lead to customer dissatisfaction. Comprehensive UV testing during development phases prevents these expensive post-market failures while extending component service life beyond standard warranty periods.
Regulatory and Quality Standards
Automotive industry standards such as SAE J2527 and ISO 4892 establish UV exposure requirements for exterior and interior components. Manufacturers must demonstrate compliance through documented testing, providing validation that materials withstand specified radiation doses. UV resistance chambers deliver the controlled, repeatable conditions necessary to meet these certification requirements across global markets.
Effects of Sunlight and UV Exposure on Plastic ComponentsSpectral Damage Characteristics
Natural sunlight contains UV radiation spanning wavelengths from 290 to 400 nanometers, with UVA (315-400nm) and UVB (280-315nm) causing distinct degradation patterns. UVB radiation, though representing smaller solar proportions, delivers higher energy that accelerates surface oxidation and color changes. UVA penetrates deeper into materials, affecting bulk properties and causing long-term mechanical property deterioration that becomes evident through embrittlement and reduced flexibility.
distinct degradation patterns. UVB radiation, though representing smaller solar proportions, delivers higher energy that accelerates surface oxidation and color changes. UVA penetrates deeper into materials, affecting bulk properties and causing long-term mechanical property deterioration that becomes evident through embrittlement and reduced flexibility.
Visual and Aesthetic Deterioration
Color shift represents one of the most noticeable effects of UV exposure on automotive plastics, and its severity is typically evaluated using a UV resistance test chamber under controlled radiation conditions. Pigments undergo photochemical breakdown, causing fading or yellowing that particularly affects dark-colored components. Surface gloss reduction occurs as microscopic crazing develops, scattering reflected light and producing a chalky appearance. These aesthetic changes diminish vehicle appeal and perceived quality regardless of maintained structural performance.
Mechanical Property Changes
Prolonged UV exposure reduces elongation at break, making plastics increasingly brittle and prone to cracking under stress. Tensile strength typically decreases by 20-40% after extended weathering, compromising structural components' load-bearing capacity. Impact resistance deteriorates significantly, causing parts that once absorbed energy during collisions to shatter unexpectedly, raising safety concerns for both occupants and pedestrians.
Simulating Real-world UV Conditions in Test ChambersAccelerated Weathering Principles
Test chambers concentrate UV radiation beyond natural sunlight intensity, compressing years of exposure into manageable testing timeframes. The UV-SI-260 model delivers controlled irradiance from 0.3 to 20 W/m², enabling researchers to establish correlation factors between laboratory hours and outdoor exposure years. This acceleration maintains relevant degradation mechanisms while eliminating the impracticality of multi-year real-time testing.
Lamp Selection and Spectral Matching
Fluorescent UV lamps provide either UVA-340 or UVB-313 spectral outputs, each simulating different environmental conditions. UVA-340 lamps replicate the critical short-wavelength portion of sunlight reaching Earth's surface, ideal for predicting outdoor durability. UVB-313 lamps deliver harsher, shorter-wavelength radiation for maximum acceleration, though this approach may produce degradation mechanisms not observed in actual service conditions.

Environmental Cycling Protocols
Realistic weathering involves more than continuous UV exposure. Chamber programs alternate between UV irradiation phases and dark condensation periods where elevated humidity attacks degraded surface layers. Temperature cycling between ambient and 90°C simulates day-night variations, while water spray systems replicate rainfall effects. These combined stresses reveal synergistic degradation that isolated testing misses.

|
Test Parameter |
Range/Specification |
Purpose |
|
UV Wavelength |
290-400nm (UVA-340 or UVB-313) |
Matches solar UV spectrum |
|
Irradiance Control |
0.3-20 W/m² |
Accelerates aging process |
|
Temperature Range |
Ambient to 90°C ±2℃ |
Simulates thermal cycling |
|
Humidity Control |
≥95% RH during condensation |
Replicates moisture exposure |
|
Specimen Distance |
50mm from lamps |
Ensures uniform irradiation |
Evaluating Mechanical and Color Stability of Automotive PlasticsTensile and Flexural Testing
Before-and-after mechanical testing quantifies UV exposure effects on load-bearing capacity. Tensile tests measure changes in ultimate strength, yield point, and elongation at break following UV resistance test chamber exposure. Flexural testing evaluates beam stiffness and failure modes under bending loads, revealing brittleness increases that threaten component functionality during normal vehicle operation and crash scenarios.
Colorimetric Analysis Methods
Spectrophotometers measure color changes using the CIELAB color space, where ΔE values quantify total color difference perception. Values below 1.0 indicate imperceptible changes, while ΔE exceeding 3.0 becomes noticeable to average observers. Automotive specifications typically require ΔE values under 2.0 after prescribed UV exposure, ensuring color consistency between adjacent components throughout vehicle life.
Surface Property Assessment
Gloss meters track surface shine degradation, with 60-degree gloss measurements providing standardized comparisons. Surface roughness profilers detect microscopic texture changes preceding visible deterioration. Fourier-transform infrared spectroscopy (FTIR) identifies chemical bond changes within surface layers, revealing oxidation products and degradation pathways before mechanical properties significantly decline, enabling predictive failure analysis.
Accelerated UV Aging Methods for Plastic PartsContinuous Exposure Protocols
Constant UV irradiation at elevated intensity provides the fastest screening method for material comparison. Sample panels mounted on chamber racks receive uniform exposure at the standardized 50mm distance, with eight 40-watt fluorescent lamps delivering consistent radiation across the test area. This approach suits preliminary material selection but may overlook temperature-humidity synergies affecting actual performance.
Cyclic Weathering Sequences
Automotive-relevant protocols alternate UV exposure with dark condensation phases, typically following SAE J2527 specifications. A common cycle includes eight hours of UVA-340 irradiation at 70°C black panel temperature, followed by four hours of condensation at 50°C with 100% relative humidity. These cycles repeat continuously for 500-2000 hours depending on application severity and performance requirements.
Water Spray Integration
Ten precision nozzles within the chamber deliver calibrated water spray during or between UV cycles, simulating rainfall and morning dew effects. The 0.5mm diameter nozzles produce fine droplets with controlled settlement rates of 0.8-1.0mm per 80cm² hourly. Water spray enhances degradation through thermal shock, physical erosion of weathered surfaces, and accelerated leaching of additives from near-surface regions.
|
Aging Method |
Typical Duration |
Best Application |
|
Continuous UV Exposure |
500-1000 hours |
Material screening and comparison |
|
Cyclic UV/Condensation |
1000-2000 hours |
Predicting outdoor durability |
|
UV with Water Spray |
1500-3000 hours |
Exterior components with rain exposure |
Predicting Long-term Durability of Automotive ComponentsCorrelation with Outdoor Weathering
Establishing relationships between chamber hours and outdoor years requires parallel exposure studies. Florida and Arizona weathering stations provide one-year reference data, while UV resistance test chamber testing compresses equivalent exposure into weeks. Correlation factors typically range from 3:1 to 8:1 depending on material, climate region, and chamber protocol, enabling manufacturers to predict five-year or ten-year performance from abbreviated laboratory testing.
Service Life Modeling Approaches
Mathematical models incorporate UV dose, temperature, and humidity data to predict time-to-failure for specific degradation modes. Arrhenius relationships describe temperature-acceleration factors, while reciprocity law principles relate radiation intensity to exposure duration. Advanced models account for non-linear degradation kinetics observed in many polymers, where initial UV exposure causes minimal change before accelerated deterioration begins.
Quality Control and Batch Verification
Production material lots undergo routine UV screening to verify consistency with qualified materials. Pass-fail criteria based on retained mechanical properties or color stability limits ensure only conforming materials enter manufacturing. Chamber testing frequency depends on supplier reliability and component criticality, ranging from every batch for safety-related parts to quarterly verification for established materials.
LIB Industry's UV Resistance Test Chamber for Reliable Temperature & Humidity ControlAdvanced Control Systems
The UV-SI-260 model features a programmable color LCD touchscreen controller with Ethernet connectivity, enabling remote monitoring and data logging throughout extended test programs. Users configure complex cycling sequences through intuitive interfaces, storing multiple protocols for different material types or industry standards. Real-time graphical displays track irradiance, temperature, and humidity parameters, alerting operators to deviations requiring attention.
logging throughout extended test programs. Users configure complex cycling sequences through intuitive interfaces, storing multiple protocols for different material types or industry standards. Real-time graphical displays track irradiance, temperature, and humidity parameters, alerting operators to deviations requiring attention.
Precision Environmental Management
Maintaining black panel temperatures between 35-80°C ensures thermal conditions match specified protocols, with ±2°C accuracy across the entire specimen plane. Automatic water supply systems and integrated purification equipment eliminate testing interruptions while preventing contamination effects on sensitive materials. The chamber achieves ≥95% relative humidity during condensation phases through precise temperature-dewpoint control.
Safety and Reliability Features
Comprehensive protection systems include over-temperature shutoff, over-current breakers, water shortage detection, earth leakage protection, and phase sequence monitoring. These redundant safeguards prevent equipment damage and ensure operator safety during unattended overnight or weekend operation. The SUS304 stainless steel interior resists corrosion from moisture and chemical emissions, maintaining chamber integrity throughout years of continuous service.
|
Control Feature |
Specification |
Benefit |
|
Temperature Control |
Ambient to 90°C ±2℃ |
Precise thermal simulation |
|
Humidity Range |
≥95% RH |
Realistic condensation conditions |
|
Irradiance Control |
0.3-20 W/m² |
Adjustable acceleration rates |
|
Programmable Cycles |
1-9999 hours, 59 minutes |
Flexible test protocols |
|
Sample Capacity |
56 pieces (75×150mm) |
High-throughput testing |
Conclusion
UV resistance testing stands as a non-negotiable requirement for automotive plastic component validation, protecting manufacturers from costly failures while ensuring vehicles maintain aesthetic appeal and structural integrity throughout their service lives. Modern UV resistance test chambers deliver precisely controlled accelerated weathering that predicts years of outdoor exposure within practical development timeframes. By implementing comprehensive UV evaluation programs using equipment like the UV-SI-260, automotive engineers make informed material selections, optimize protective treatments, and validate component designs against demanding industry standards.
FAQWhat duration of UV chamber testing equals one year of outdoor exposure?
The correlation varies by material and climate, typically ranging from 500-1500 chamber hours equaling one outdoor year. Florida subtropical conditions require different correlation factors than Arizona desert environments, while polymer chemistry significantly affects degradation rates under accelerated testing.
Can UV resistance chambers test assembled components or only flat samples?
Chambers accommodate both approaches. The UV-SI-260 provides flat specimen holders for standardized material coupons plus 3D holders for testing assembled parts like mirror housings, bumper sections, or complete dashboard assemblies, revealing stress-concentration effects not evident in flat-sample testing.
How do UVA-340 and UVB-313 lamps differ in automotive testing applications?
UVA-340 lamps replicate the critical short-wavelength portion of natural sunlight, providing realistic outdoor weathering simulation. UVB-313 lamps deliver more aggressive shorter wavelengths for maximum acceleration but may produce degradation mechanisms not observed in actual vehicle service, making them better suited for quality control screening.
What maintenance requirements ensure accurate UV chamber performance?
Lamp replacement follows manufacturer-specified intervals based on operating hours, typically 1000-2000 hours depending on intensity settings. Irradiance sensors require annual calibration, while water system filters need quarterly replacement. Regular cleaning of interior surfaces prevents contamination effects on sensitive test specimens.
LIB Industry, a leading UV resistance test chamber manufacturer and supplier, specializes in turn-key environmental testing solutions tailored to automotive applications. Contact our technical team at ellen@lib-industry.com to discuss your specific UV testing requirements and explore how our advanced chamber technology can strengthen your quality assurance programs.
Hot and Cold Chamber Testing for Drug Temperature & Humidity Control
One year after purchasing our product, Rob, a procurement manager at an electronics company, conducted a temperature and humidity calibration and shared his satisfaction: 'Although we haven't been using the chamber frequently lately, I believe it's still performing well.' Let's take a closer look at the Thermal Shock Chamber.

Pharmaceutical stability hinges on precise environmental control during storage and distribution. Hot and cold chamber testing validates drug formulations against temperature fluctuations and varying humidity levels, ensuring active ingredients maintain potency throughout their shelf life. These specialized chambers replicate extreme conditions - from arctic cold to tropical heat - allowing manufacturers to predict degradation patterns, optimize packaging, and meet stringent ICH stability guidelines. By subjecting medications to controlled stress scenarios, pharmaceutical companies safeguard patient safety while demonstrating regulatory compliance across global markets.
Why Temperature and Humidity Control is Critical in Drug Storage?
Chemical Degradation Mechanisms in Pharmaceuticals
Temperature variations trigger hydrolysis, oxidation, and photodegradation reactions that compromise drug efficacy. Aspirin, when exposed to moisture above 60% RH, breaks down into salicylic acid and acetic acid, producing a characteristic vinegar odor. Protein-based biologics denature at elevated temperatures, losing their three-dimensional structure and therapeutic properties. Heat accelerates molecular movement, increasing collision frequency between reactive species and catalyzing unwanted chemical transformations.
moisture above 60% RH, breaks down into salicylic acid and acetic acid, producing a characteristic vinegar odor. Protein-based biologics denature at elevated temperatures, losing their three-dimensional structure and therapeutic properties. Heat accelerates molecular movement, increasing collision frequency between reactive species and catalyzing unwanted chemical transformations.
Moisture-Induced Physical Changes
Humidity directly impacts tablet hardness, dissolution rates, and powder flowability. Hygroscopic excipients absorb atmospheric moisture, causing caking in granules or softening in capsules. Moisture infiltration promotes microbial proliferation in liquid formulations, compromising sterility. Lyophilized products require storage below 25% RH to prevent reconstitution before use, while some sustained-release coatings lose integrity when exposed to fluctuating humidity levels.
Patient Safety and Treatment Efficacy
Subpotent medications deliver insufficient therapeutic doses, prolonging illness or allowing disease progression. Degraded compounds may generate toxic byproducts - penicillin breakdown produces allergenic substances that trigger hypersensitivity reactions. Temperature excursions during shipping to tropical regions have caused insulin analog precipitation, rendering injections ineffective for diabetic patients. Stringent environmental control protects vulnerable populations from treatment failures and adverse reactions.
Key Parameters in Hot and Cold Chamber Drug Testing
Temperature Range Selection and Specifications
|
Temperature Zone |
Application |
Typical Range |
|
Refrigerated Storage |
Vaccines, Biologics |
2°C to 8°C |
|
Controlled Room Temperature |
Tablets, Capsules |
15°C to 25°C |
|
Accelerated Testing |
Stability Prediction |
40°C to 50°C |
|
Extreme Stress Testing |
Shipping Validation |
-40°C to 60°C |
Modern hot and cold chambers achieve temperature fluctuation within ±0.5°C and deviation below ±2.0°C, matching pharmaceutical stability protocol requirements. The 1°C/min cooling rate and 3°C/min heating rate enable controlled thermal transitions without shocking sensitive formulations. French TECUMSEH compressors deliver reliable performance across -70°C to +150°C spectrum, accommodating both cryogenic preservation studies and heat-resistant packaging evaluations.
Humidity Control and Monitoring Systems
Relative humidity parameters span 20% to 98% RH with ±2.5% deviation tolerance. External isolation humidifiers prevent cross-contamination between samples while stainless steel surface evaporation technology ensures uniform moisture distribution. Automatic water purification systems eliminate mineral deposits that could interfere with humidity sensors. Programmable controllers synchronize temperature and humidity profiles, creating complex environmental cycles that mirror seasonal variations or transportation routes.
Calibration and Validation Requirements
Chambers undergo quarterly calibration using NIST-traceable sensors positioned throughout the working volume. Mapping studies identify hot spots and cold zones, verifying spatial uniformity. Temperature probes placed at geometric centers and corners confirm that all sample locations experience identical conditions. Humidity verification employs saturated salt solutions as reference standards. Documentation packages include calibration certificates, validation protocols, and equipment qualification reports that satisfy FDA 21 CFR Part 11 electronic record requirements.
Simulating Real-world Environmental Conditions for Pharmaceuticals
Geographic Climate Zone Replication
Pharmaceutical products destined for equatorial markets undergo testing at 30°C/75% RH, representing tropical storage conditions per ICH Zone IV guidelines. Mediterranean climates require validation at 25°C/60% RH, while temperate regions use 21°C/45% RH baseline conditions. Chambers programmed with diurnal temperature cycling - mimicking day-night fluctuations - reveal photo-stability issues masked by constant-condition testing. Arctic distribution channels demand freeze-thaw cycling between -20°C and 5°C to assess emulsion separation or crystallization in suspensions.
guidelines. Mediterranean climates require validation at 25°C/60% RH, while temperate regions use 21°C/45% RH baseline conditions. Chambers programmed with diurnal temperature cycling - mimicking day-night fluctuations - reveal photo-stability issues masked by constant-condition testing. Arctic distribution channels demand freeze-thaw cycling between -20°C and 5°C to assess emulsion separation or crystallization in suspensions.
Transportation Stress Simulation
|
Transport Mode |
Temperature Challenge |
Duration |
|
Air Cargo |
-40°C to 50°C swings |
24-72 hours |
|
Ocean Shipping |
15°C to 35°C gradients |
2-6 weeks |
|
Desert Ground Transport |
60°C peak exposure |
8-12 hours |
Chambers replicate cargo hold conditions where medications experience rapid altitude-induced pressure changes combined with temperature extremes. Shake tables integrated with environmental chambers simultaneously apply vibration stress, revealing synergistic degradation effects. Thermal shock protocols alternate between -20°C and 40°C every 6 hours, identifying packaging failures before products reach pharmacy shelves.
Seasonal Variation Modeling
Annual stability programs incorporate chambers cycling through spring humidity spikes, summer heat peaks, autumn dryness, and winter cold. This approach captures cumulative degradation that single-condition testing overlooks. Pharmaceutical warehouses in continental climates may see 40°C summer temperatures drop to -10°C in winter - conditions that hot and cold chambers reproduce across compressed timescales. Seasonal modeling reveals whether protective packaging maintains internal microclimates when external environments fluctuate dramatically.

Accelerated Stability Testing for Drug Formulations
Arrhenius Equation Application in Shelf-Life Prediction
Elevated temperature testing exploits the Arrhenius relationship where reaction rates double with every 10°C increase. Storing samples at 40°C/75% RH for six months generates degradation data equivalent to two years at 25°C, enabling faster market entry. Pharmaceutical scientists plot degradation kinetics at multiple temperatures, extrapolating shelf-life predictions through regression analysis. This mathematical modeling requires validation through real-time studies but provides preliminary stability estimates within months instead of years.
Forced Degradation Study Protocols
|
Stress Condition |
Target Parameter |
Analytical Method |
|
60°C Heat |
Thermal Decomposition |
HPLC Assay |
|
80% RH Humidity |
Moisture Uptake |
Karl Fischer |
|
Light Exposure |
Photodegradation |
UV-Vis Spectroscopy |
Chambers subject formulations to 50°C/75% RH for three months, identifying potential degradation pathways and impurity profiles. Oxidative stress chambers incorporate peroxide challenge or oxygen-enriched atmospheres. These aggressive conditions reveal formulation weaknesses - inadequate antioxidants, permeable packaging, or unstable excipient combinations. Degradation products undergo structural characterization, informing quality specifications and analytical method development.
Statistical Analysis and Confidence Intervals
Stability data requires statistical rigor to support regulatory submissions. Multiple chamber units running parallel studies provide replication necessary for meaningful variance calculations. Regression analysis determines degradation rates with 95% confidence intervals. Outlier detection algorithms identify aberrant data points caused by equipment malfunction or sampling errors. Statistical software packages generate shelf-life estimates with upper and lower bounds, accounting for batch-to-batch variability and analytical uncertainty.
Evaluating Active Ingredient Potency under Stress
HPLC Quantification of Degraded Compounds
High-performance liquid chromatography separates parent drug from degradation products, quantifying potency loss over time. Samples withdrawn at predetermined intervals undergo dissolution testing followed by chromatographic analysis. Method validation confirms linearity across 80% to 120% of labeled claim, with precision below 2% RSD. Stability-indicating methods distinguish between active pharmaceutical ingredient and structurally related impurities, meeting USP <1225> requirements.
Physical Property Assessment Techniques
Beyond chemical assay, hot and cold chambers enable monitoring of dissolution profiles, hardness evolution, and friability changes. Tablets stored at high humidity may soften, affecting disintegration times. Capsules become brittle under dry conditions, cracking during handling. Appearance attributes - color shifts, coating defects, or phase separation - signal formulation instability. Texture analyzers quantify mechanical strength degradation, correlating environmental exposure with compromised quality attributes.
Bioavailability Implications of Degradation
Potency reduction below 90% of labeled claim renders medications subtherapeutic. Crystalline form conversions alter dissolution kinetics, changing absorption rates in the gastrointestinal tract. Enantiomeric purity shifts may produce inactive or toxic stereoisomers. Accelerated testing identifies these critical quality attributes before distribution, preventing therapeutic failures. Bioequivalence studies sometimes repeat with aged samples to confirm pharmacokinetic equivalence throughout product shelf life.
Ensuring Regulatory Compliance in Drug Storage
ICH Stability Guideline Implementation
ICH Q1A(R2) mandates long-term testing at 25°C/60% RH for 12 months minimum, with accelerated studies at 40°C/75% RH for six months. Chambers meeting these specifications generate data acceptable to FDA, EMA, and WHO regulatory bodies. Intermediate conditions at 30°C/65% RH bridge gaps between standard protocols. Chambers equipped with data logging systems create audit trails documenting every temperature excursion, humidity deviation, or power interruption - critical evidence during regulatory inspections.
Good Manufacturing Practice Alignment
Environmental chambers reside within validated facilities maintaining ISO 14644 cleanroom classifications where appropriate. Standard operating procedures govern chamber loading, sample handling, and condition monitoring. Change control processes document any modifications to testing protocols or equipment parameters. Annual preventive maintenance includes gasket replacement, sensor recalibration, and refrigerant system evaluation. Deviation investigations analyze root causes when chamber performance drifts outside acceptance criteria.
Documentation and Traceability Systems
Electronic data capture systems timestamp all measurements, linking chamber performance to specific product batches. Programmable controllers generate reports showing temperature-humidity profiles throughout study duration. Chain-of-custody records track samples from manufacturing through stability chamber to analytical laboratory. Regulatory submissions include chamber qualification documents, calibration certificates, and deviation reports. This comprehensive documentation satisfies 21 CFR Part 211 requirements for pharmaceutical manufacturing facilities.
Ensure Pharmaceutical Stability with LIB Industry's Precise Hot and Cold Chamber
Advanced Technical Specifications
LIB Industry hot and cold chambers feature 304 stainless steel mirror-finish interiors resistant to pharmaceutical compound corrosion. Double- layer tempered glass observation windows with 8cm insulation enable visual monitoring without thermal compromise. Programmable color LCD touchscreen controllers with Ethernet connectivity facilitate remote monitoring and data export. Polyurethane foam insulation maintains thermal efficiency while nichrome heaters provide rapid, uniform temperature distribution. Mechanical compression refrigeration using French TECUMSEH compressors ensures reliable operation across demanding pharmaceutical testing protocols.
layer tempered glass observation windows with 8cm insulation enable visual monitoring without thermal compromise. Programmable color LCD touchscreen controllers with Ethernet connectivity facilitate remote monitoring and data export. Polyurethane foam insulation maintains thermal efficiency while nichrome heaters provide rapid, uniform temperature distribution. Mechanical compression refrigeration using French TECUMSEH compressors ensures reliable operation across demanding pharmaceutical testing protocols.
Comprehensive Safety and Protection Features
Multi-layer safety systems include humidifier dry-combustion protection, over-temperature shutoff, and refrigerant high-pressure alarms. Water shortage sensors prevent humidifier damage while earth leakage protection safeguards personnel. Over-current protection circuits defend against electrical faults. These redundant safety mechanisms minimize downtime and protect valuable pharmaceutical samples from environmental excursions. Automatic water supply systems with purification modules eliminate manual intervention, reducing contamination risks during extended stability studies.
Customization and Service Excellence
LIB Industry provides turnkey solutions encompassing chamber design, installation, commissioning, and operator training. Standard configurations include cable ports with sealing plugs and adjustable shelving, with custom options for specialized sample fixtures. Models ranging from 100L benchtop units to 1000L walk-in chambers accommodate diverse testing volumes. Three-year warranties demonstrate manufacturing confidence while door-to-door delivery service ensures global accessibility. Technical support teams assist with protocol development, troubleshooting, and regulatory compliance questions throughout the product lifecycle.
Conclusion
Hot and cold chamber testing forms the foundation of pharmaceutical quality assurance, validating drug stability across diverse environmental challenges. Precise temperature and humidity control enables accelerated shelf-life predictions, regulatory compliance demonstration, and patient safety protection. Advanced chamber technology replicates real-world stress conditions, revealing formulation vulnerabilities before market distribution. Statistical rigor applied to stability data generates defensible shelf-life claims accepted by global regulatory authorities.
FAQsWhat temperature range is required for ICH-compliant pharmaceutical stability testing?
ICH guidelines specify long-term storage at 25°C±2°C with 60%±5% RH, while accelerated testing requires 40°C±2°C at 75%±5% RH. Chambers must maintain these conditions with minimal fluctuation, typically ±0.5°C temperature stability and ±2.5% humidity deviation to generate regulatory-acceptable data.
How do hot and cold chambers prevent cross-contamination between different drug samples?
Modern chambers utilize external isolation humidification systems and stainless steel construction to minimize contamination risks. Separate shelving, proper spacing between samples, and regular chamber cleaning protocols prevent compound migration. Some facilities dedicate specific chambers to particular drug classes, eliminating cross-contamination concerns entirely.
Can accelerated stability testing completely replace real-time studies for pharmaceutical products?
Accelerated testing predicts shelf life through mathematical modeling but cannot fully replace real-time studies. Regulatory agencies require ongoing real-time data to confirm accelerated predictions and detect long-term degradation pathways that elevated temperatures might not reveal. Both testing approaches complement each other in comprehensive stability programs.
Ready to elevate your pharmaceutical stability testing program? LIB Industry, a leading hot and cold chamber manufacturer and supplier, delivers precision environmental testing solutions tailored to your drug development needs. Contact our technical team at ellen@lib-industry.com to discuss custom chamber configurations and validation support.
Dust Proof Test Chamber Applications in PCB and Component Testing
Modern electronics face relentless challenges from airborne particulates that threaten operational integrity. Printed circuit boards (PCBs) and electronic components require rigorous validation against dust infiltration to guarantee performance across diverse environments. A dust proof test chamber replicates severe conditions where fine particles penetrate enclosures, accumulate on sensitive surfaces, and compromise electrical pathways. This specialized equipment enables manufacturers to identify vulnerabilities early, refine protective measures, and deliver products that withstand industrial, automotive, and outdoor applications. Through controlled exposure to standardized dust concentrations, engineers obtain quantifiable data about sealing effectiveness, material resilience, and long-term reliability before market deployment.
Why Dust Testing is Critical for PCB Reliability?Understanding Contamination Risks
Microscopic particles infiltrate electronic assemblies through ventilation openings, seams, and connector interfaces. Once inside, dust accumulates on solder joints, component leads, and PCB traces, creating conductive bridges that trigger short circuits or increase leakage currents. Environmental test chambers simulate these scenarios by circulating calibrated dust concentrations, exposing assemblies to conditions mirroring years of field deployment within controlled timeframes.
accumulates on solder joints, component leads, and PCB traces, creating conductive bridges that trigger short circuits or increase leakage currents. Environmental test chambers simulate these scenarios by circulating calibrated dust concentrations, exposing assemblies to conditions mirroring years of field deployment within controlled timeframes.
Preventing Field Failures
Products deployed without adequate dust testing face premature failure rates that damage brand reputation and inflate warranty costs. Automotive electronics, industrial controls, and telecommunications equipment operate in environments where silica, metal particles, and organic debris constantly threaten system stability. Validation through dust proof test chambers identifies weak points in enclosure design, allowing engineering teams to implement corrective measures before production scales.
Regulatory Compliance Requirements
International standards mandate dust resistance verification for equipment operating in harsh environments. Ingress Protection (IP) ratings specify the level of protection against solid particle intrusion, with IP5X and IP6X designations requiring specialized testing. Test chambers meeting IEC 60529 and ISO 20653 specifications provide the controlled conditions necessary to certify products, ensuring market access across regulated industries and geographic regions.
Key Dust Proof Test Standards for Electronic ComponentsIEC 60529 Testing Protocol
This international standard defines dust test procedures using talcum powder with particles sized 50 micrometers in diameter. Test specimens remain in chambers maintaining 2-4 kg/m³ dust concentration for prescribed durations, typically eight hours with continuous circulation. Evaluation criteria examine whether dust deposits interfere with safe operation or accumulate in quantities that could compromise protection ratings over the product lifecycle.
|
|
|
| Name | dust proof test chamber |
|
Workroom dimension (mm) |
800*800*800 D*W*H |
|
External dimension (mm) |
950*3150*1800 D*W*H |
|
Interior Volume (L) |
510 |
|
Diameter of Turntable (mm) |
600 |
|
Turntable loads |
20kgs Max |
|
Turntable Rotation Speed |
0~7r/min (Adjustable) |
|
Internal Diameter of IPX5 Nozzle |
6.3 mm |
|
Internal Diameter of IPX6 Nozzle |
12.5 mm |
|
Water Flow Rate IPX5/ IPX6 |
12.5L/min ±5% / 100L/min ±5% |
|
Controller |
Programmable color LCD touch screen controller |
|
Ethernet connection, PC Link, USB |
|
|
Build-in Water Tank(mm) |
370*375*950 |
|
View Window Size(mm) |
475*475 |
ISO 20653 Automotive Applications
Automotive electronics require enhanced testing protocols addressing vehicle-specific contamination scenarios. ISO 20653 extends IEC 60529 requirements with modified dust compositions and environmental conditions reflecting road dust, industrial pollutants, and climatic variations. Dust proof test chambers configured for this standard maintain precise temperature ranges from ambient to 50°C while controlling humidity levels below 30% RH, replicating dashboard installations and under-hood environments.
MIL-STD-810 Military Standards
Defense and aerospace applications demand extreme environmental validation. MIL-STD-810 Method 510 specifies blowing dust and sand tests using Arizona road dust with particle distributions matching desert conditions. Test chambers must generate wind velocities exceeding 18 meters per second while maintaining dust concentrations and exposure durations that stress enclosures beyond commercial requirements, revealing failure modes that only emerge under severe operational stresses.
|
Standard |
Particle Type |
Concentration |
Test Duration |
Application Sector |
|
IEC 60529 |
Talcum powder (50μm) |
2-4 kg/m³ |
8 hours |
Consumer electronics, industrial equipment |
|
ISO 20653 |
Road dust mixture |
2-4 kg/m³ |
Variable by rating |
Automotive electronics, transportation |
|
MIL-STD-810 |
Arizona road dust |
High velocity exposure |
6+ hours |
Military, aerospace, defense systems |
Simulating Harsh Environments for PCBsTemperature-Controlled Dust Exposure
Real-world electronics encounter simultaneous thermal and particulate stresses. Advanced test chambers integrate nichrome heating elements that elevate internal temperatures to 50°C while maintaining uniform dust circulation. This combination reveals whether thermal expansion compromises sealing integrity or whether elevated temperatures alter particle adhesion characteristics, providing comprehensive validation beyond single-stress testing approaches.
that elevate internal temperatures to 50°C while maintaining uniform dust circulation. This combination reveals whether thermal expansion compromises sealing integrity or whether elevated temperatures alter particle adhesion characteristics, providing comprehensive validation beyond single-stress testing approaches.
Pressure Differential Testing
Ventilated electronics generate internal pressure variations that drive dust infiltration through microscopic gaps. Chambers equipped with vacuum systems and pressure regulation simulate these dynamics by cycling between positive and negative pressure states. The resulting particle penetration patterns identify sealing vulnerabilities that only manifest when equipment operates with active cooling systems or undergoes altitude changes during transportation.
Observation Window Capabilities
Large glass observation ports with integrated dust scrapers enable real-time monitoring throughout test cycles. Engineers document particle accumulation patterns, assess filter loading rates, and identify unexpected entry pathways without interrupting controlled conditions. This visual feedback accelerates design iteration by providing immediate evidence of protective measure effectiveness or revealing failure mechanisms requiring investigation.
Monitoring Component Performance under Dust ExposureElectrical Continuity Verification
Test specimens connect to external monitoring equipment through dust-proof power outlets rated for 16A capacity. Continuous measurement of voltage, current, and resistance parameters throughout exposure cycles detects degradation trends before catastrophic failures occur. Subtle increases in contact resistance or leakage currents signal progressive contamination, enabling engineers to establish maintenance intervals and cleaning protocols for fielded equipment.
Functional Operation Assessment
Active operation during dust testing reveals functional impairments that escape detection during passive exposure. Rotating components, cooling fans, and mechanical switches undergo accelerated wear when contaminated, producing performance degradation data relevant to lifecycle predictions. Dust proof test chambers supporting specimen power delivery enable comprehensive validation where electronics operate normally while dust circulates, replicating actual service conditions.

Post-Test Inspection Protocols
Following exposure cycles, disassembly and microscopic examination quantify particle penetration depth and distribution patterns. Weighing assemblies before and after testing establishes mass gain metrics correlating with dust ingress severity. Combining gravimetric measurements with visual documentation creates objective pass/fail criteria aligned with product specifications, supporting quality assurance decisions backed by reproducible evidence.
|
Measurement Parameter |
Monitoring Method |
Acceptance Criteria |
Failure Indication |
|
Electrical resistance |
Continuous ohmmeter readings |
<5% deviation from baseline |
>10% increase suggests contamination |
|
Functional operation |
Active device testing |
All features operational |
Intermittent faults or degraded performance |
|
Mass gain |
Precision weighing |
<0.5g increase per 1000cm² |
Excessive accumulation indicating poor sealing |
Material and Design Considerations for Dust ResistanceGasket Selection Strategies
Sealing materials must maintain compression across temperature ranges while resisting abrasive particle erosion. Silicone gaskets offer temperature stability, but closed-cell foam provides superior dust barrier properties for low-pressure applications. Test chamber validation compares competing materials under identical conditions, generating empirical data that informs material selection decisions rather than relying solely on supplier specifications.
Enclosure Geometry Optimization
Labyrinth seals, overlapping joints, and baffle designs create tortuous pathways that reduce particle penetration probability. Computer simulations predict theoretical performance, but dust chamber testing validates actual effectiveness when exposed to turbulent airflow and particle impact forces. Iterative testing of prototype designs identifies optimal configurations balancing manufacturing complexity against protection performance requirements.
Surface Treatment Effects
Conformal coatings, anodizing, and specialized finishes alter particle adhesion characteristics on PCB assemblies and enclosure surfaces. Chamber testing with coated and uncoated specimens quantifies protection improvements, justifying additional processing costs through measurable reliability enhancements. Comparative studies reveal which treatments provide adequate protection versus those delivering marginal benefits insufficient to warrant production implementation.
Predicting Long-term PCB StabilityAccelerated Lifecycle Testing
Condensing years of environmental exposure into concentrated test cycles requires elevated stress levels that accelerate degradation mechanisms without introducing unrealistic failure modes. Chambers maintaining 2-4 kg/m³ concentrations far exceed typical ambient conditions, compressing timeline predictions when correlated with field data. Statistical models translate chamber hours into equivalent operational years, supporting warranty calculations and maintenance scheduling.
Correlation with Field Data
Validation programs compare dust proof test chamber predictions against actual product performance in deployed environments. Telemetry from fielded electronics provides contamination accumulation rates, failure distributions, and maintenance intervention requirements. Aligning test protocols with real-world outcomes refines acceleration factors and ensures chamber testing accurately represents service life expectations across varied deployment scenarios.
Establishing Maintenance Intervals
Dust testing data informs service documentation specifying cleaning frequencies and filter replacement schedules. Quantifying performance degradation rates versus particle exposure enables condition-based maintenance strategies that optimize operational costs while preventing premature failures. Chamber testing establishes thresholds where intervention becomes necessary, replacing arbitrary time-based maintenance with evidence-driven schedules.
|
Test Duration |
Equivalent Field Exposure |
Typical Assessment |
Application Example |
|
8 hours |
1-2 years moderate environment |
Standard IP rating verification |
Indoor industrial controls |
|
24 hours |
3-5 years harsh environment |
Extended reliability validation |
Outdoor telecommunications |
|
72 hours |
10+ years extreme conditions |
Military/aerospace qualification |
Defense systems, aviation |
Ensure Electronics Reliability with LIB Industry's Advanced Dust Proof Test ChamberProgrammable Control Capabilities
Modern test chambers feature color LCD touchscreen controllers with Ethernet connectivity, enabling remote monitoring and automated test sequence execution. Programmable parameters include temperature ramping profiles, dust circulation intervals, and pressure cycling patterns that replicate specific deployment scenarios. PC connectivity facilitates data logging and report generation, streamlining compliance documentation and design validation workflows.
Precision Environmental Control
Maintaining stable test conditions requires sophisticated climate management systems. Chambers regulate temperature within ±2°C across the working volume while humidity control prevents condensation that would alter particle characteristics. Pressure gauges, air filters, and regulators ensure reproducible conditions between test runs, eliminating variability that could compromise comparative studies or regulatory submissions.
Robust Construction Standards
Test equipment endures continuous operation with abrasive materials circulating at high velocities. Chambers constructed from 304 stainless steel interior surfaces resist corrosion and particle embedding, while cold-rolled steel exteriors with electrostatic powder coating withstand industrial environments. Electromagnetic door locks prevent accidental opening during test cycles, protecting personnel and maintaining chamber integrity throughout demanding validation programs.
Versatile Configuration Options
Multiple chamber sizes accommodate components ranging from individual PCBs to complete electronic enclosures. Models offering 800-liter and 1000-liter internal volumes provide flexibility for testing small assemblies or full-scale products. Caster-mounted designs with foot brakes enable laboratory reconfiguration without permanent installation, adapting test facilities to evolving project requirements and production schedules.
Conclusion
Dust proof test chambers deliver indispensable validation for electronics facing particulate contamination challenges. Through standardized protocols aligned with international requirements, these specialized tools quantify protective measure effectiveness, predict long-term reliability, and prevent costly field failures. Comprehensive testing programs combining environmental stress, electrical monitoring, and material evaluation ensure products meet performance expectations across demanding applications where dust exposure threatens operational integrity and customer satisfaction.
FAQsWhat dust particle size is used in standard PCB testing?
Standard protocols utilize talcum powder with 50-micrometer diameter particles and 75-micrometer wire gap spacing, as specified by IEC 60529. This particle size represents typical environmental contaminants that penetrate enclosures and accumulate on electronic assemblies, providing realistic validation of protective measures against common industrial and commercial dust exposures.
How long should PCBs remain in dust chambers during testing?
Typical test durations range from eight hours for basic IP rating verification to 72 hours for military and aerospace qualifications. Testing duration depends on target protection level, deployment environment severity, and regulatory requirements. Accelerated testing condenses years of field exposure into concentrated cycles through elevated dust concentrations exceeding ambient conditions.
Can dust testing damage electronic components permanently?
Properly configured test protocols minimize component damage risk while revealing design vulnerabilities. Non-powered testing poses minimal risk, whereas active operation testing may accelerate wear on mechanical components. Post-test cleaning removes accumulated particles, allowing engineers to implement design improvements before production. Testing identifies weaknesses requiring correction rather than causing irreparable damage to validated assemblies.
Partner with LIB Industry for Comprehensive Testing Solutions
LIB Industry manufactures advanced dust proof test chambers engineered to international standards, providing manufacturers and suppliers with reliable qualification equipment. Our turn-key solutions encompass research, design, production, commissioning, and training tailored to your specific testing requirements. Contact our technical team at ellen@lib-industry.com to discuss how our dust testing capabilities support your electronics reliability objectives and accelerate product development timelines.
Shelf Life Test Chamber for Food Packaging Stability Testing
Ensuring food packaging performs reliably throughout its intended shelf life is critical to product quality, safety, and regulatory compliance. A shelf life test chamber enables manufacturers to evaluate packaging stability under precisely controlled temperature and humidity conditions that simulate real storage, transportation, and retail environments. By identifying material degradation, seal failures, and barrier performance changes early in development, companies can validate packaging designs with confidence before products reach the market.
A shelf life test chamber serves as an essential instrument for evaluating how food packaging performs under controlled environmental conditions over extended periods. These specialized chambers simulate various temperature and humidity scenarios that products might encounter during storage, transportation, and retail display. By accelerating aging processes through precise climate control, manufacturers can predict packaging degradation, identify potential failure points, and validate barrier properties before products reach consumers. This testing approach helps ensure that food packaging maintains its protective qualities throughout the intended shelf life, safeguarding product quality, consumer safety, and brand reputation.
What Factors Affect Food Packaging Stability?
Environmental Stress Variables
Food packaging faces multiple environmental challenges that impact its protective capabilities. Temperature extremes can cause materials to expand, contract, or degrade at varying rates. Polymers may become brittle in cold conditions while adhesives might soften under heat. Humidity levels directly influence moisture barrier performance, affecting seal integrity and material dimensional stability.
expand, contract, or degrade at varying rates. Polymers may become brittle in cold conditions while adhesives might soften under heat. Humidity levels directly influence moisture barrier performance, affecting seal integrity and material dimensional stability.
Chemical Interactions and Migration
Packaging materials can undergo chemical changes when exposed to certain food components or atmospheric gases. Oxygen permeation rates affect oxidative rancidity in fats and oils. Volatile organic compounds from packaging may migrate into food products, potentially altering taste profiles or raising safety concerns about compliance with regulatory standards.
Mechanical Degradation Pathways
Physical stresses accumulate during handling, stacking, and transportation. Flexural fatigue weakens seal areas while compression forces test structural integrity. Repeated thermal cycling creates stress points where cracks may initiate. Understanding these mechanical degradation patterns helps designers create more resilient packaging solutions.
Role of Temperature and Humidity in Shelf Life TestingPrecise Climate Control Requirements
Accurate environmental simulation demands tight control over temperature fluctuation and humidity distribution. Advanced shelf life test chambers maintain stability within ±0.5°C and carefully regulate moisture levels from 20% to 98% RH. This precision ensures reproducible results that accurately reflect real-world storage conditions rather than introducing variability from equipment limitations.
Thermal Cycling Protocols
Repeated temperature transitions reveal weaknesses that steady-state conditions might miss. Cycling between extreme temperatures stresses seal interfaces, tests adhesive bonds, and exposes material incompatibilities. Heating rates of 3°C/min and controlled cooling enable realistic simulation of daily temperature variations or seasonal changes.
Humidity Impact Assessment
Moisture significantly affects packaging performance across multiple dimensions. High humidity challenges barrier films, tests seal integrity against water vapor transmission, and accelerates corrosion of metal components. Low humidity conditions assess brittleness and static electricity generation in protective films.
|
Parameter |
TH-225 Model |
TH-500 Model |
|
Internal Dimensions |
500×600×750 mm |
700×800×900 mm |
|
Temperature Range |
-86°C to +150°C |
-86°C to +150°C |
|
Humidity Range |
20%-98% RH |
20%-98% RH |
|
Temperature Deviation |
±2.0°C |
±2.0°C |
Accelerated Aging for Food ProductsTime Compression Methodology
Accelerated aging applies elevated stress levels to compress months or years of natural aging into weeks of testing. The Arrhenius equation provides mathematical relationships between temperature elevation and reaction rate acceleration. This scientific approach enables rapid validation of packaging performance predictions without waiting for real-time aging completion.
Q10 Factor Applications
The Q10 relationship describes how reaction rates typically double with every 10°C temperature increase. Packaging engineers use this principle to calculate equivalent aging periods at elevated temperatures. Testing at 40°C for one month might simulate six months at ambient conditions, depending on specific material characteristics.
Validation Against Real-Time Data
Accelerated testing requires correlation with actual shelf life observations to ensure accuracy. Manufacturers typically run parallel studies comparing accelerated results with real-time aging samples. This validation confirms that elevated stress conditions produce degradation mechanisms similar to natural aging rather than introducing artificial failure modes.
Monitoring Packaging Integrity over Time
| Name | shelf life test chamber | |||||
|
Model |
TH-100 |
|||||
|
Internal dimension (mm) |
400*500*500 |
|||||
|
Overall dimension (mm) |
860*1050*1620 |
|||||
|
Capacity |
100L |
|||||
|
Temperature range |
-20℃ ~+150 ℃ |
|||||
|
Low type |
A: -40℃ B:-70℃ C -86℃ |
|||||
|
Humidity Range |
20%-98%RH |
|||||
|
Temperature deviation |
± 2.0 ℃ |
|||||
|
Heating rate |
3 ℃ / min |
|||||
|
Cooling rate |
1 ℃ / min |
|||||
|
Controller |
Programmable color LCD touch screen controller, Multi-language interface, Ethernet , USB |
|||||
|
Exterior material |
Steel Plate with protective coating |
|||||
|
Interior material |
SUS304 stainless steel |
|||||
|
Standard configuration |
1 Cable hole (Φ 50) with plug; 2 shelves |
|||||
|
Timing Function |
0.1~999.9 (S,M,H) settable |
|||||
Visual Inspection Protocols
Regular examination reveals surface changes, discoloration, delamination, or physical deformation. Trained observers document seal appearance, material transparency, and any visible defects. Photographic documentation creates objective records for comparison across testing intervals and between different packaging configurations.
Instrumental Measurement Techniques
Quantitative assessments provide objective data about packaging performance changes when samples are evaluated in a shelf life test chamber. Tensile testing measures mechanical strength degradation while gas chromatography detects permeability increases. Seal strength analyzers evaluate bond integrity at multiple time points throughout accelerated aging cycles.
Barrier Property Evaluation
Oxygen transmission rates, water vapor permeability, and aromatic compound retention define packaging protective capabilities. Specialized instruments measure these parameters before and after environmental exposure. Degradation patterns help predict when barrier properties might fall below acceptable thresholds during actual distribution.
|
Test Parameter |
Measurement Method |
Acceptance Criteria |
|
Seal Strength |
Tensile Testing |
>15 N/15mm width |
|
Oxygen Transmission |
Gas Chromatography |
<50 cc/m²/day |
|
Water Vapor Permeability |
Gravimetric Method |
<5 g/m²/day |
Impact of Material Selection on Product Shelf LifePolymer Compatibility Considerations
Different plastic resins exhibit unique responses to environmental stresses. Polyethylene maintains flexibility across wide temperature ranges while polypropylene offers superior moisture barriers. Polyester provides excellent oxygen resistance but may become brittle under UV exposure. Matching material properties to product requirements optimizes protective performance.
Multi-Layer Structure Advantages
Combining materials with complementary properties creates superior barriers. Aluminum foil layers block light and provide exceptional oxygen barriers. Ethylene vinyl alcohol copolymers enhance gas resistance while polyethylene layers ensure heat sealability. Adhesive layers must maintain cohesion throughout temperature and humidity cycling.

Sustainable Material Challenges
Bio-based and compostable packaging materials present unique testing challenges. These materials may degrade intentionally under certain conditions while maintaining protection during intended shelf life. Chamber testing validates that degradation occurs only under appropriate composting conditions rather than during normal storage.
Predictive Analysis for Food Product QualityStatistical Modeling Approaches
Regression analysis correlates environmental exposure with packaging performance metrics. Weibull distributions predict failure probabilities across product populations. These statistical tools transform shelf life test chamber test data into actionable shelf life predictions with defined confidence intervals.
across product populations. These statistical tools transform shelf life test chamber test data into actionable shelf life predictions with defined confidence intervals.
Arrhenius Kinetics Application
Temperature-dependent degradation rates follow predictable mathematical relationships. Plotting degradation data against inverse absolute temperature yields activation energies for specific failure mechanisms. These calculations enable extrapolation from accelerated conditions to estimate performance at lower storage temperatures.
Risk Assessment Integration
Combining packaging degradation data with microbiological growth models provides comprehensive safety predictions. Understanding how barrier failure correlates with pathogen growth or toxin formation helps establish conservative shelf life limits. This integrated approach balances quality maintenance with consumer safety priorities.
|
Storage Condition |
Predicted Shelf Life |
Confidence Level |
|
-18°C (Frozen) |
24 months |
95% |
|
4°C (Refrigerated) |
90 days |
95% |
|
25°C (Ambient) |
12 months |
90% |
Ensure Food Safety & Quality with LIB Industry's Precise Shelf Life Test ChamberAdvanced Climate Control Technology
LIB chambers incorporate R404A and R23 refrigerants for ultra-low temperature capabilities reaching -86°C. Programmable touchscreen controllers with multi-language interfaces simplify complex testing protocols. Forced air convection systems distribute temperature uniformly throughout the testing space, eliminating hot spots that could compromise data integrity.
throughout the testing space, eliminating hot spots that could compromise data integrity.
Customizable Testing Solutions
Beyond standard configurations, LIB offers specialized modifications including explosion-proof designs for volatile product testing, low-pressure simulation for high-altitude distribution scenarios, and integrated vibration platforms for transportation simulation. PT100Ω temperature sensors and precision humidity probes ensure measurement accuracy throughout extended testing campaigns.
Remote Monitoring Capabilities
Network connectivity enables real-time data access via Ethernet or laboratory Wi-Fi systems. Researchers can monitor ongoing tests, receive alerts about parameter deviations, and download results through USB interfaces. This connectivity supports multi-site collaboration and enables continuous oversight without constant physical presence.
Maintenance and Cleaning Efficiency
Mirror-finish stainless steel interiors resist corrosion while simplifying sanitation after tests involving food spoilage. Perforated shelving allows drainage and air circulation while supporting diverse sample configurations. Separate water and electrical systems enhance operational safety during cleaning procedures and routine maintenance activities.
Conclusion
Shelf life test chambers provide critical validation tools for ensuring food packaging maintains protective qualities throughout distribution cycles. Through precise environmental control, accelerated aging protocols, and comprehensive monitoring approaches, manufacturers gain confidence that their packaging solutions will perform reliably. Advanced chamber technology from specialized manufacturers enables thorough evaluation of material performance, seal integrity, and barrier properties under conditions that accurately simulate real-world challenges.
FAQsHow long does accelerated shelf life testing typically take?
Accelerated testing duration depends on target shelf life and acceleration factors applied. Most food packaging studies require 3-12 weeks of chamber testing to simulate 6-24 months of real-time aging, with validation studies running parallel real-time controls.
Can shelf life chambers test multiple packaging formats simultaneously?
Modern chambers accommodate diverse sample configurations on adjustable shelving systems. Researchers can test pouches, bottles, trays, and cartons within the same run, provided all samples require identical environmental conditions and testing protocols throughout the study period.
What maintenance requirements do shelf life test chambers have?
Regular maintenance includes refrigerant level verification, sensor calibration verification, cleaning of condensate drains, and inspection of door seals. Most manufacturers recommend professional calibration annually, with routine cleaning after each test cycle involving potentially contaminating samples.
Partner with LIB Industry for Your Testing Needs
As a leading shelf life test chamber manufacturer and supplier, LIB Environmental Simulation Industry delivers precision equipment backed by comprehensive technical support. Our factory produces CE-approved chambers designed for demanding food packaging applications. Contact our team at ellen@lib-industry.com to discuss your specific testing requirements and receive detailed quotations for standard or custom configurations.
- 1
- 16
- 17
- 18
- 78